Team studies mechanism of H. influenzae biofilm formation
A research study identifying novel bacterial physiology in the creation of biofilms by Nationwide Children's Hospital scientists has been published in the current issue of Proceedings of the National Academy of Sciences (PNAS).
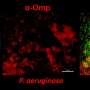
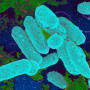
The laboratory of Lauren Bakaletz, PhD, director, Center for Microbial Pathogenesis and vice president of basic sciences, The Research Institute at Nationwide Children's, studied the biofilm construction capabilities of nontypeable Haemophilus influenzae (NTHI), a bacterium responsible for sinusitis, pneumonia, exacerbations of cystic fibrosis and COPD, bronchitis and ear infections.
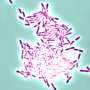
Biofilms are large 3D communities of bacteria that adhere to body surfaces and protect bacteria from environmental stressors such as antibiotics and antibodies. The lab found that when H. influenzae builds its biofilms, it does so via an active and regulated means while remaining intact, unlike other bacteria which self-sacrifice in order to contribute to the biofilm. Other types of bacteria either explode, sending their DNA into the biofilm, or shuttle their DNA out into the environment through a syringe-like appendage produced by the bacterium, all to benefit the potency of the biofilm.
The novel mechanism by which nontypeable H. influenzae releases DNA through an inner membrane pore complex that partners with another complex in the outer membrane allows DNA to be ejected out into the biofilm, via a previously unidentified process.
"We're very excited that our work with biofilms has been accepted for publication by the Proceedings of the National Academy of Sciences," says Dr. Bakaletz. "Our lab hopes to use what we've learned about H. influenzae biofilms to identify vaccine targets as well as improve existing methods of therapeutic treatment for the diseases of the respiratory tract caused by this prevalent pathogen."
More information: Joseph A. Jurcisek et al, Nontypeable Haemophilus influenzae releases DNA and DNABII proteins via a T4SS-like complex and ComE of the type IV pilus machinery, Proceedings of the National Academy of Sciences (2017). DOI: 10.1073/pnas.1705508114
Journal information: Proceedings of the National Academy of Sciences
Provided by Nationwide Children's Hospital