X-ray photoelectron spectroscopy under real ambient pressure conditions
Researchers at Institute for Molecular Science (IMS), Innovation Research Center for Fuel Cells, University of Electro-Communications, Research Center for Materials Science, Nagoya University, and JASRI (Japan Synchrotron Radiation Research Institute), have improved an ambient-pressure photoelectron spectroscopy instrument using hard X-rays produced at SPring-8 and succeeded in photoelectron spectrometry under real atmospheric pressure for the first time in the world. Their achievements has been published online in the Applied Physics Express.
Conventional photoelectron spectroscopy can only measure samples under high vacuum, while many catalytic reactions occur under atmospheric pressure. The discrepancy between the findings obtained by experiments under high vacuum and the actual reaction mechanism under atmospheric pressure, "pressure gap," has been an issue. In recent years, in order to fill this gap, an apparatus called "ambient pressure photoelectron spectroscopy" has been developed that enables measurement under gas atmosphere. However, the upper-pressure limit of operation in a general ambient pressure photoelectron spectrometer is approximately 5,000 Pa. Even the apparatus with a currently reported world's highest performance has a limit of 15,000 Pa (approx. 0.15 atm), which is about 1/7 the atmospheric pressure (approx. 100,000 Pa). Therefore, various research groups in the world have been working on the development of photoelectron spectroscopy that operate under higher gas pressure.
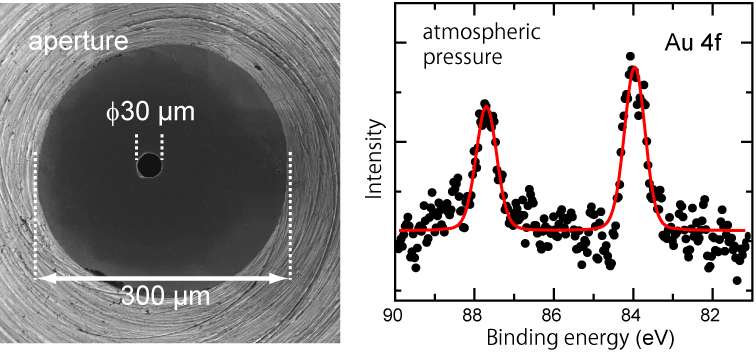
A problem upon measurement using ambient pressure photoelectron spectrometer is "energy decay" of the photoelectrons emitted from the sample exposed to light, which is due to scattering caused by gas. This limits the upper-pressure of the measurement. "We made two improvements," explains Yasumasa Takagi, an assistant professor of IMS. "First, we used hard X-rays that has higher energy compared to soft X-rays and boosted kinetic energy of the photoelectrons. Next, we created an extremely tiny aperture of 30 μm in diameter (figure left), which is a port that accepts photoelectrons into the spectrometer. This enabled to shorten the distance between the sample and the aperture, i.e. the distance of photoelectron traveling through gas has shortened." Thus, using gold thin film as a sample, the research group succeeded in photoelectron spectroscopy under real atmospheric pressure, for the first time in the world (figure right).
Professor Toshihiko Yokoyama (IMS) has a vision of possibilities for future applications of the novel photoelectron spectrometer. "Our apparatus achieved photoelectron spectroscopy under real atmospheric pressure, which greatly broadened its range of application. Reactions between solid and gas such as catalytic reactions and electrode reactions in fuel cells can be directly examined under atmospheric pressure. It can be also applied to biological samples that are fragile under high vacuum. In the future, photoelectron spectroscopy will be used for state analysis in various research areas."
Provided by National Institutes of Natural Sciences