New sensors could enable more affordable detection of pollution and diseases
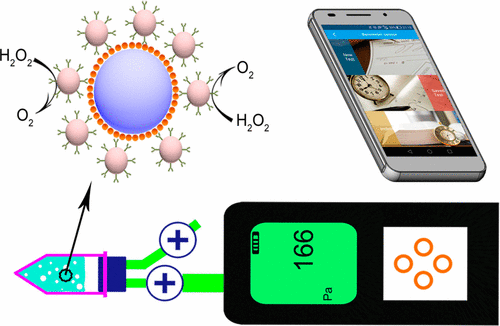
When it comes to testing for cancer, environmental pollution and food contaminants, traditional sensors can help. The challenges are that they often are bulky, expensive, non-intuitive and complicated. Now, one team reports in ACS Sensors that portable pressure-based detectors coupled with smartphone software could provide a simpler, more affordable alternative while still maintaining sensitivity.
Current disease and contamination sensors require expensive readout equipment or trained personnel. Yuehe Lin, Yong Tang and colleagues propose a new detection system based on pressure changes. For example, when a disease biomarker is present, it causes a chain reaction in the device that results in oxygen being released and pressure building. The pressure changes are measured by a portable barometer, and smartphone software provides an easy readout of the results.
To show the versatility of the pressure sensor, the team tested a variety of applications. Prototypes could detect carcinoembryonic antigen, a protein present in high levels in patients with colon or rectal cancer; ractopamine, which is an animal-feed additive banned in many countries; and thrombin, a cardiovascular disease marker. In addition, a mercury-ion sensor was developed for environmental pollution monitoring. The researchers say that because the results are immediately available with a smartphone, the method could enable real-time monitoring of environmental pollution, disease outbreaks and food safety.
More information: Qiangqiang Fu et al. Versatile Barometer Biosensor Based on Au@Pt Core/Shell Nanoparticle Probe, ACS Sensors (2017). DOI: 10.1021/acssensors.7b00156
Abstract
There is a high global demand for sensitive, portable, user-friendly, and cost-effective biosensors. In this work, we introduce a barometer-based biosensor for the detection of a broad range of targets. The device is operated by measuring the pressure change produced by oxygen (O2) generation in a limited chamber using a portable barometer. The design employs core–shell Au@Pt nanoparticles (Au@PtNPs) as the bioassay probe to catalyze the decomposition of H2O2 and the release of O2. As a proof of concept, we developed barometer-based immunosensors to detect carcinoembryonic antigen (CEA) and ractopamine (Rac). In addition, barometer-based aptasensors for sensitive detection of thrombin and mercury ion (Hg2+) were also developed. In order to facilitate the analysis of results, we have developed smartphone software to calculate, save, and wirelessly trsnsmit the results. Linear detection ranges for detection of CEA, Rac, thrombin, and Hg2+ were 0.025–1.6 ng/mL, 0.0625–4 ng/mL, 4–128 U/L, and 0.25–16 ng/mL, respectively. The detection limit of these four analytes is 0.021 ng/mL, 0.051 ng/mL, 2.4 U/L, and 0.22 ng/mL, respectively. Furthermore, the developed barometer-based biosensors exhibited high specificities for these four analytes. CEA in serum samples, Rac in urine samples, thrombin in serum samples, and Hg2+ in river water samples were measured by the barometer-based biosensors. Obtained results of these targets from barometer-based biosensors were consistent with detection results from traditional methods, indicating that barometer-based biosensors are widely applicable.
Provided by American Chemical Society


















