New insight into a central biological dogma on ion transport
New research results from Aarhus University and New York University show how active transport of potassium can be achieved by a membrane protein complex that has roots in both ion pump and ion channel super-families. The results, which have just been published in Nature, shed new light on what define channels and pumps.
Potassium is highly concentrated in all cells, and it is the main determinant of turgor pressure. Too much potassium and the cell can rupture due to water influx, too little and the cell will shrivel as water leaves. This osmotic principle is used also in food preservation, when salting or drying meats, fruits and vegetables.
As potassium is virtually impermeable to the cell membrane, potassium transporters have evolved that will mediate its uptake. In unicellular organisms, the mechanisms to maintain internal potassium levels are highly responsive due to the challenges a fluctuating environment can present. Normally, potassium channels will create inward flux of potassium driven by the electrochemical gradient. However, in environments with very little potassium present, this potential cannot maintain inward flux, and active transport through potassium pump systems rapidly takes over to directly drive the uptake of potassium.
The mechanism and structure of these potassium pump systems are not well understood, and from sequence analysis they appear to have elements pertaining both to potassium channels and to more classical sodium/potassium pumps known from animals. This has puzzled researchers for decades, and many almost even doubted the existence of these complexes as a functional unit.
This is, in part, because of how ion transport is taught in biology as a concept of 'channels versus pumps'. Ion channels mediate fast and passive 'downhill' transport, while ion pumps mediate slow and active 'uphill' transport against an electrochemical gradient. Both processes are relatively well understood, and are normally juxtapositioned as completely different entities.
Channel and pump super-families create active ion transport together
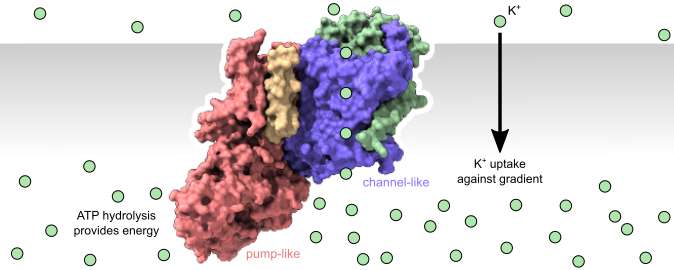
Results published this week in Nature challenge this comfortable separation and address the puzzling question of how pumps and channels might work together in a complex. For the first time, a joint research collaboration between Aarhus University and New York University have visualized a potassium transporting structure where members from channel and pump super-families come together in a large complex to create active transport. One 'pump-like' subunit creates the energy input (similar to a motor), while a 'channel-like' subunit has been re-purposed to function as an active carrier mediating transport of potassium against a 10,000 fold gradient.
"To me the most fascinating part of this whole thing is that it is a break with the boxes we normally like to put our biological concepts in," says AIAS fellow and Assistant Professor Bjørn Panyella Pedersen, part of the small team that has published the results. "It is yet another fascinating example that nature doesn't care about our attempts to make sense of it and classify it. If evolution can make it work, it happens."
The results build on a strong foundation of prior knowledge of channel and pump function. The concept of gated channels has been known and studied for a long time, and indeed most ion channels have one or more gates as an essential part of their physiological function. Similarly, the molecular mechanism behind ion pump function is well described. With the new results we can suddenly see how gates from a channel-like subunit can be linked up to a pump-like subunit that can sequentially control their opening and closing. This creates a strong coupling between gate opening/closing, substrate recognition/binding and energy expenditure. These are all the hallmarks of active transport known from other pump systems, but here they only work if all elements of the complex are together.
"This has been one of the hardest, but also most rewarding structures I have ever had the pleasure of working on. So many new ideas have come out of this, and the interpretation of the results was challenging as we really needed to think outside the box", Bjørn says with a grin, before continuing, "David [Prof. David Stokes] was the key to this, as he has worked on the complex for many years and has an extensive knowledge of the intricacies of the system. This was a true team effort, and we will continue our collaboration to further our knowledge of what makes this complex tick, on the basis of the ideas we have now."
More information: Ching-Shin Huang et al, Crystal structure of the potassium-importing KdpFABC membrane complex, Nature (2017). DOI: 10.1038/nature22970
Journal information: Nature
Provided by Aarhus University