Facile decomposition of metal-NHC complexes under catalytic conditions and the key role of NHC-stabilized nanoparticles
Metal complexes bearing N-heterocyclic carbene ligands (NHC ligands) have been successfully used in a number of catalytic reactions. High stability of metal-NHC complexes and convenient synthesis of well-defined catalyst precursors are valuable advantages for practical applications. Strong metal-ligand binding and high stability of metal-NHC complexes under catalytic conditions have been widely assumed.
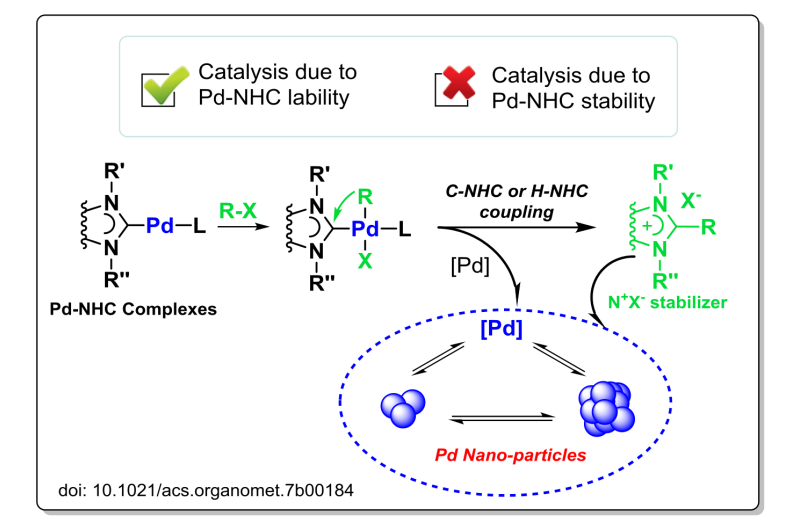
However, a recent study has shown that Pd-NHC complexes readily release NHC ligands into solution and generate Pd clusters and nanoparticles. After the first step of the catalytic reaction (oxidative addition of organic halide, R-X), the palladium complexes turn to the R-Pd(NHC)-X species. Surprisingly, R-NHC coupling involving the NHC ligand and the organic group R readily occurs and leads to the dissociation of NHC ligand framework to solution. The experimental study has detected the formation of metal nanoparticles, and a series of tests has confirmed the key role of the nanoparticles in the catalytic reaction. Quantum chemical calculations have found a relatively small activation barrier for the R-NHC coupling, which makes the process readily accessible under regular reaction conditions.
Indeed, the study has shown that NHC ligands can be easily released into solution in the form of azolium salts due to H-NHC or C-NHC couplings. The azolium salts, formed from the dissociated NHC ligands, act as stabilizers and play an important role in stabilization of metal nanoparticles in solution. The study has revealed a new mode of operation of Pd-NHC systems, where catalysis is governed by the lability of the Pd-NHC framework, rather than its previously assumed stability.
The importance of the findings is far beyond the studied Mizoroki-Heck reaction. The mechanism of several catalytic reactions may be reinvestigated to assess the impact of the lability of metal-NHC complexes.
More information: Alexander V. Astakhov et al, A New Mode of Operation of Pd-NHC Systems Studied in a Catalytic Mizoroki–Heck Reaction, Organometallics (2017). DOI: 10.1021/acs.organomet.7b00184
Provided by Zelinsky Institute of Organic Chemistry