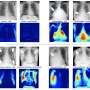
Machine learning models for drug discovery

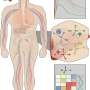
IBM today announced that its scientists have been granted a patent on machine learning models to predict therapeutic indications and side effects from various drug information sources. IBM Research has implemented a cognitive association engine to identify significant linkages between predicted therapeutic indications and side effects, and a visual analytics system to support the interactive exploration of these associations.
This approach could help researchers in pharmaceutical companies to generate hypotheses for drug discovery. For instance, strongly correlated disease-side-effect pairs identified by the patented invention could be beneficial for drug discovery in many ways. One could use the side-effect information to repurpose existing treatments (e.g. drugs causing postural hypotension could be potential candidates for treating hypertension). If a new drug is being designed for a disease that is strongly correlated with severe side effects, then special attention could be paid to controlling the formulation and dosing of the drug in the clinical trials to prevent serious safety issues.
IBM was granted U.S. Patent 9,536,194: Method and system for exploring the associations between drug side-effects and therapeutic indications for this invention.
Lack of efficacy and adverse side effects are two of the primary reasons a drug fails clinical trials, each accounting for around 30 percent of failures. Computational models and machine learning methods that can derive useful insights from large amounts of data on drugs and diseases from various sources hold great promise for reducing these attrition rates and improving the drug discovery process.
"As inventors at IBM, we have the opportunity to help solve real-world problems," said Jianying Hu, Senior Manager and Program Director, Center for Computational Health, IBM Research. "Our team is dedicated to this research and we continue to search for new ways to improve people's health around the world through innovation and invention."
One of the research areas of the Center for Computational Health at IBM Thomas J. Watson Research Center is translational informatics, which focuses on the development of novel techniques to extract insights and knowledge from biological and clinical data to support biological scientists, clinicians, and patients.
IBM has been working in this area for several years, and has developed a suite of advanced machine learning tools as well as computational models and platforms that can be used to derive insights from a wide variety of data sources (e.g. pharmacological knowledge bases) to help improve the efficiency and effectiveness of drug discovery and development. These methodologies have been applied to many specific use cases including drug repurposing (i.e. helping to find new uses for existing drugs in the market), indication expansion (i.e. helping to identify potential new indications for drugs still in various stages of a development pipeline), drug safety (i.e. helping to detect and predict a drug's safety profiles) and personalized medicine (e.g. personalized efficacy/safety profile predictions).
More information: patft.uspto.gov/netacgi/nph-Pa … patentnumber=9536194
Provided by IBM