One synthetic molecule, two doorways into cell

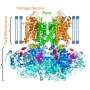
Shuhei Furukawa from Kyoto University's Institute for Integrated Cell-Material Sciences (iCeMS) and colleagues in Japan synthesised polyhedral-shaped porous molecules that rotate in a double layer of lipids, exposing different-shaped openings. These openings allowed the molecule to conduct ions from one side of the artificial lipid membrane to the other in two distinct ways. Chemists had previously synthesised molecules that conduct ions across a lipid membrane, but incorporating two different openings into a single molecule remained elusive.
Ion channels are porous proteins present in the membranes of all living cells. They are responsible for managing the transportation of ions across the cell membrane, creating electrical signals that initiate various cellular processes. Most ion channels have a single opening, but a unique family of channels have two. Researchers would like to have a clearer understanding of how these two-pored channels work and whether they might be implicated in some diseases. Designing synthetic channels is one way to go about this.
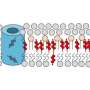
Furukawa and his team synthesised a molecule made of the chemical element rhodium. The molecule is shaped like a cuboctahedron, with eight triangular and six square faces. The rhodium-based molecule was embedded into a double layer of lipids. The team then synthesized short and long alkoxy chains made of hydrogen, carbon and oxygen molecules, which attached the molecule to the surrounding lipids.
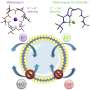
They found that the rhodium-based molecule rotated inside the lipid bilayer, exposing at any one time a smaller triangular- or a larger square-shaped channel. Each shape led to a different state of electrical conductance. The molecule was also found to rotate more slowly when it was attached to the longer alkoxy chains, increasing the amount of time the channel remained open before the molecule continued its rotation.
"Rotational dynamics is the key to switching between distinct conductance states to expose either the triangular or square apertures to the aqueous phase," write the researchers in their study published in the journal Chem.
The team also found that changing the metal they used from rhodium to copper changed the molecule's permeability: while the rhodium-based molecule is permeable to ions with two positive charges, like calcium, the copper-based molecule only transports ions with a single positive charge, like potassium.
Further studies are underway to improve the molecule's design so the pores are open more often and the molecules can be switched from one state to the other using external stimuli.
More information: Ryuji Kawano et al, Metal-Organic Cuboctahedra for Synthetic Ion Channels with Multiple Conductance States, Chem (2017). DOI: 10.1016/j.chempr.2017.02.002
Provided by Kyoto University