Synbio for bioremediation—fighting plastic pollution

Plastic pollution is one of the major problems of our times. Plastic is a durable material that can be moulded into solid objects of the most disparate shapes. Properties such as low cost, versatility and easy manufacturing are behind its overwhelming success. So much so that in just over a century – the first plastic, bakelite, was invented in NY in 1907- the world and, in particular, our oceans have become clogged by it.
Plastic is non-biodegradable, and it is estimated that 8 million metric tonnes of plastic pollution enter the ocean each year.
To give a more tangible idea, our oceans are now filled with 'the equivalent of 5 grocery bags full of plastic trash on every 30 cm of every nation's coastline around the globe.'
The major contributor is China, which produces circa 30% of the total global of plastic waste, with the USA and Europe being also in the list of the top 20 producers. The consequences are devastating to the sea life (I recommend this article about the effects of microplastics in our oceans, and the documentary cited within).
Apart from the obvious need of spreading the word so that we can all become more socially aware and use less and less plastic, another solution to the problem could be found in bioremediation. Bioremediation is defined as the use of either naturally occurring or deliberately introduced microorganisms or other forms of life to consume and break down environmental pollutants, in order to clean up a polluted site.
PET (polyethylene terephthalate) is a widely used plastic. Even though it is the most recycled plastic, only about 30% of it is, in fact, recycled in the States (50% in Europe). The rest of it is disposed of in landfills (I also recommend this map that shows how much of the US has become covered in landfills since the first of these sites was created in 1937). Until now, the use of microorganisms for PET disposal or recycling had not been considered a viable alternative as bioremediation had not yet succeeded in delivering a feasible process – there were only few reports on microorganisms able to degrade PEG and these were limited to a few fungal species.
At the beginning of 2016, however, a group of Japanese researchers identified a novel bacterium, Ideonella sakaiensis 201-F6, that shows great promises as an alternative to the disposal of PET. The bacterium appears to be able to fully degrade the plastic, by feeding on it as its major energy and carbon source.
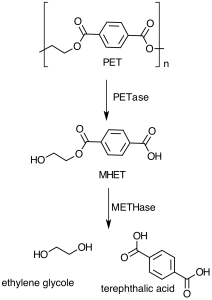
Ideonella sakaiensis catalyses the degradation of PET thanks to the production of two hydrolytic enzymes, which the researchers called PETase and MHETase.
The PET plastic is metabolised into its monomers terephtalic acid and ethylene glycole, molecules that are generally derived from raw petroleum. Hence, PET degradation could not only be used as a strategy for environmental remediation, but also as a platform for the recycling of PET waste products, such as terephtalic acid, that could then be used to generate more plastic, without the need of using up more petroleum.
The research
Shosuke Yoshida and colleagues collected and screened 250 PET contaminated samples from a PET bottle recycling site in order to individuate microorganisms that could feed on low-crystallinity PET film. Among the samples, they isolated Ideonella sakaiensis 201-F. In particular, the bacterium demonstrated to be able to almost completely degrade the PET film after 6 weeks at 30 °C.
The next step was that of identifying the enzyme responsible for this acquired ability of Ideonella to hydrolyze the plastic. Upon further testing, the researchers discovered two hydrolases that work in tandem to disassemble the plastic polymer, which they named PETase and MHETase. PETase acts first to break down PET to mono(2-hydroxyehyl)terephthalic acid (MHET). MHET is then subsequently hydrolyzed by the METHase into ethylene glycole and terephtalic acid, which the bacterium uses for growth.
A very interesting point made by Prof. Bornscheuer in his Science Perspective article (Feeding on plastics) is that the enzymes PETase and METHase appear to be evolutionary unique, in what they are very different from any enzyme encountered until now that showed somewhat ability to hydrolyze ester bonds in PET. Their role in the sakaiensis strain appears to be solely the degradation of the PET polymer, which could also imply that these enzymes have evolved their peculiar activities very rapidly since PET has been around only for the past 70 years.
It will be very interesting to see how this research progresses both from an applied point of view, and to shed light on the evolutionary history behind the two new hydrolases. Furthermore, this study certainly sets the ground for the potential discovery of similar microorganisms with degrading capabilities for different uses.
More information: S. Yoshida et al. A bacterium that degrades and assimilates poly(ethylene terephthalate), Science (2016). DOI: 10.1126/science.aad6359
Journal information: Science
Provided by PLOS Blogs
This story is republished courtesy of PLOS Blogs: blogs.plos.org.





















