Credit: American Chemical Society
In the 1960s, the introduction of fabric softeners transformed rough, scratchy clothes into softer, more comfortable garments. But recently, the products' popularity has dipped in part due to millennials' concern for their potential environmental impact, according to recent news reports. Now one team has developed a better method for evaluating fabric softeners that could lead to potentially "greener"—and more millennial-friendly—versions. Their study appears in ACS' The Journal of Physical Chemistry B.
Fabric softeners' main ingredients are surfactants, which are compounds that stick to clothing during the rinse cycle and make them feel softer. Scientists have been studying fabric softeners for decades, and many believe that surfactants work by forming a lubricating layer on clothing. But to date, no one knows how the softeners work on a molecular level. So, Evdokia K. Oikonomou, Jean-François Berret and colleagues set out to devise a new method to look more closely at the interaction between cotton fabric and fabric softeners as a first step toward developing more eco-friendly products.
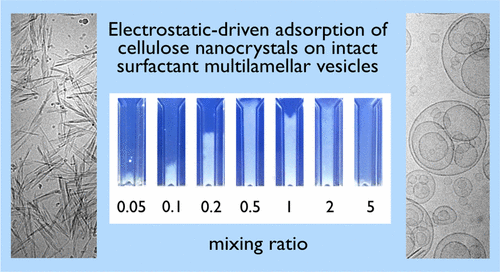
The researchers studied how a double-tailed cationic surfactant, which is often used in commercial fabric softeners, would combine with natural cellulose nanocrystals—a stand-in for cotton—using multiple techniques including light scattering and optical and electron microscopy. The study showed that the concentration of surfactant affected vesicle size. The concentration of surfactant also affected how these vesicles arranged themselves, sometimes nesting within each other to form multiple layers. The researchers say their technique could speed up manufacturers' assessment of fabric softeners' efficiency and potentially help get newer, more environmentally friendly products to the market faster.
More information: E. K. Oikonomou et al. Fabric Softener–Cellulose Nanocrystal Interaction: A Model for Assessing Surfactant Deposition on Cotton, The Journal of Physical Chemistry B (2017). DOI: 10.1021/acs.jpcb.7b00191
Abstract
There is currently a renewed interest for improving household and personal-care formulations to provide more environment-friendly products. Fabric conditioners used as softeners have to fulfill a number of stability and biodegradability requirements. They should also display significant adsorption on cotton under the conditions of use. The quantification of surfactant adsorption remains however difficult because the fabric-woven structure is complex and deposited amounts are generally small. Here, we propose a method to evaluate cellulose–surfactant interactions with increased detection sensitivity. The method is based on the use of cellulose nanocrystals (CNCs) in lieu of micron-sized fibers or yarns, combined with different techniques, including light scattering, optical and electron microscopy, and electrophoretic mobility. CNCs are rod-shaped nanoparticles in the form of 200 nm laths that are negatively charged and can be dispersed in bulk solutions. In this work, we use a double-tailed cationic surfactant present in fabric softener. Results show that the surfactants self-assemble into unilamellar, multivesicular, and multilamellar vesicles, and the interaction with CNCs is driven by electrostatics. Mutual interactions are strong and lead to the formation of large-scale aggregates, where the vesicles remain intact at the cellulose surface. The technique developed here could be exploited to rapidly assess the fabric conditioner efficiency obtained by varying the nature and content of their chemical additives.
Journal information: Journal of Physical Chemistry B
Provided by American Chemical Society