Biochemical 'fossil' shows how life may have emerged without phosphate
One major mystery about life's origin is how phosphate became an essential building block of genetic and metabolic machinery in cells, given its poor accessibility on early Earth. In a study published on March 9 in the journal Cell, researchers used systems biology approaches to tackle this long-standing conundrum, providing compelling, data-driven evidence that primitive life forms may not have relied on phosphate at all. Instead, a few simple, abundant molecules could have supported the emergence of a sulfur-based, phosphate-free metabolism, which expanded to form a rich network of biochemical reactions capable of supporting the synthesis of a broad category of key biomolecules.
"The significance of this work is that future efforts to understand life's origin should take into account the concrete possibility that phosphate-based processes, which are essential today, may not have been around when the first life-like processes started emerging," says senior study author Daniel Segrè of Boston University. "An early phosphate-independent metabolism capable of producing several key building blocks of living systems is in principle viable."
Phosphate is essential for all living systems and is present in a large proportion of known biomolecules. A sugar-phosphate backbone forms the structural framework of nucleic acids, including DNA and RNA. Moreover, phosphate is a critical component of adenosine triphosphate (ATP), which transports chemical energy within cells, and a compound called NADH, which has several essential roles in metabolism. But it is unclear how phosphate could have assumed these central roles on primordial Earth, given its scarcity and poor accessibility.
In light of this puzzle, some have proposed that early metabolic pathways did not rely on phosphate. In many of these scenarios, sulfur and iron found on mineral surfaces are thought to have fulfilled major catalytic and energetic functions prior to the appearance of phosphate. One notable origin-of-life scenario suggests that the role of ATP was originally assumed by sulfur-containing compounds called thioesters, which are widely involved in protein, carbohydrate, and lipid metabolism. Despite the availability of iron and sulfur on early Earth, concrete evidence supporting these scenarios has been lacking.
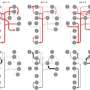
To test the feasibility of the "iron-sulfur world hypothesis" and the "thioester world scenario," Segrè and his team used computational systems biology approaches originally developed for large-scale analyses of complex metabolic networks. The researchers used a large database to assemble the complete set of all known biochemical reactions. After exploring this so-called "biosphere-level metabolism," the researchers identified a set of eight phosphate-free compounds thought to have been available in prebiotic environments. They then used an algorithm that simulated the emergence of primitive metabolic networks by compiling all possible reactions that could have taken place in the presence of these eight compounds, which included formate, acetate, hydrogen sulfide, ammonium, carbon dioxide, water, bicarbonate, and nitrogen gas.
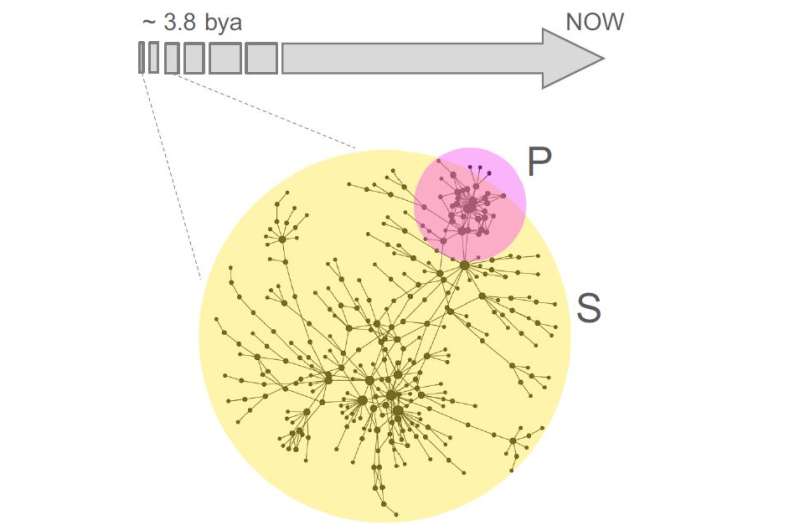
This analysis revealed that a few simple prebiotic compounds could support the emergence of a rich, phosphate-independent metabolic network. This core network, consisting of 315 reactions and 260 metabolites, was capable of supporting the biosynthesis of a broad category of key biomolecules such as amino acids and carboxylic acids. Notably, the network was enriched for enzymes containing iron-sulfur clusters, bolstering the idea that modern biochemistry emerged from mineral geochemistry. Moreover, thioesters rather than phosphate could have enabled this core metabolism to overcome energetic bottlenecks and expand under physiologically realistic conditions.
"Before our study, other researchers had proposed a sulfur-based early biochemistry, with hints that phosphate may not have been necessary until later," Segrè says. "What was missing until now was data-driven evidence that these early processes, rather than scattered reactions, could have constituted a highly connected and relatively rich primitive metabolic network."
Although this non-experimental evidence does not definitively prove that life started without phosphate, it provides compelling support for the iron-sulfur world hypothesis and the thioester world scenario. At the same time, the study calls into question the "RNA world hypothesis," which proposes that self-replicating RNA molecules were the precursors to all current life on Earth. Instead, the results support the "metabolism-first hypothesis," which posits that a self-sustaining phosphate-free metabolic network predated the emergence of nucleic acids. In other words, nucleic acids could have been an outcome of early evolutionary processes rather than a prerequisite for them.
"Evidence that an early metabolism could have functioned without phosphate indicates that phosphate may have not been an essential ingredient for the onset of cellular life," says first author Joshua Goldford of Boston University. "This proto-metabolic system would have required an energy source and may have emerged either on the Earth's surface, with solar energy as the main driving force, or in the depth of the oceans near hydrothermal vents, where geochemical gradients could have driven the first life-like processes."
In future studies, the researchers will continue to apply systems biology approaches to study the origin of life. "My hope is that these findings will motivate further studies of the landscape of possible historical paths of metabolism, as well as specific experiments for testing the feasibility of a phosphate-free sulfur-based core biochemistry," Segrè says. "The idea of analyzing metabolism as an ecosystem-level or even planetary phenomenon, rather than an organism-specific one, may also have implications for our understanding of microbial communities. Furthermore, it will be interesting to revisit the question of how inheritance and evolution could have worked prior to the appearance of biopolymers."
More information: Cell, Goldford et al: "Remnants of an Ancient Metabolism without Phosphate" www.cell.com/cell/fulltext/S0092-8674(17)30133-2 , DOI: 10.1016/j.cell.2017.02.001
Journal information: Cell
Provided by Cell Press