When batteries explode: five questions

Samsung is warning consumers to stop using its flagship Galaxy Note 7 smartphone after a spate of battery explosions.
It was the latest in a string of lithium-ion battery fires on products ranging from laptops to hoverboards to airliners—and a reminder that pushing the technology envelope can sometimes be problematic.
Here are some things to know about the recall and why batteries can be a fire hazard.
How do these batteries work and why do they catch fire?
A lithium-ion battery is a kind of rechargeable battery that uses different materials, one holding positive ions—the cathode—and the other holding negative ions—the anode.
These ions move one way when charging, and back again when discharging—being used.
These two layers—or conductors—are never supposed to touch, so manufacturers insert separators to keep them apart.
Unfortunately, the chemical reaction that makes batteries work also creates heat. Overcharging the packs—or charging them too fast—can lead to fires.
What is the issue with Samsung's batteries?
We don't know exactly, but Samsung has given some clues. It said parts of the battery that should never touch came together due to a "very rare manufacturing process error".
Gadget makers weigh all sorts of factors like performance, cost and safety when rolling out their next-generation technology.
And the race to push more battery life into their latest phone or tablet can lead to unexpected results.
"Smartphone makers are trying to squeeze these batteries into a small, thin package," said Hideki Yasuda, an analyst at Ace Research Institute in Tokyo.
"Since batteries generate energy through a chemical reaction, it's really hard to reduce the risk (of fire) to zero.
"Sometimes convenience comes with a price."
Has this happened before?
Yes. Battery fires have been reported in products including Sony Vaio laptops, last-year's must-have toy the hoverboard, electric bikes and even Boeing's Dreamliner jet.
In Samsung's case, faulty batteries caused some handsets to burst into flames during charging.
Cases of exploding batteries continued to emerge following the recall announcement, as users claimed a burning device had damaged a hotel room in Australia or set a car on fire in the US.
How extensive is the problem?
Not very. Millions of lithium-ion batteries are produced annually with a tiny fraction of those catching fire or exploding.
At the start of this month, Samsung had received reports of about 35 incidents involving the phone's batteries.
"It's not easy to know if Samsung's problem is the same as others...at this point," Yasuda said.
"If its battery suppliers sell these same ones to other producers,it could possibly affect them too."
What does this mean for Samsung and other gadget makers that use these batteries?
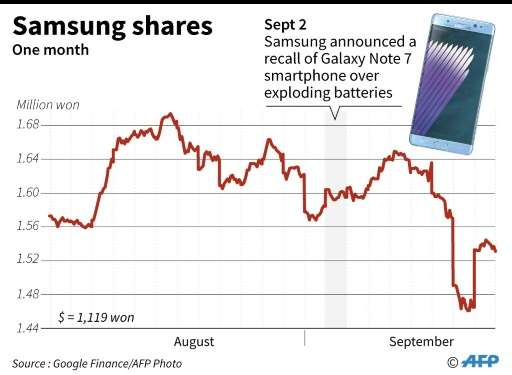
It will damage Samsung's reputation and likely hurt profits at a time when it is feeling the squeeze from Apple's iPhone and Chinese rivals in the lower-end category. The mobile business accounts for a major share of Samsung's profits.
It will also put pressure on gadget makers to figure out how to make battery packs safe for ever-smaller devices.
"We always want batteries to be safe but also to be more efficient," said Guy Marlair, a France-based safety expert.
"The more that we boost the battery's performance, the higher the energy density in a small space and the tougher it is to manage security."
© 2016 AFP