Researchers outsmart the biological uncertainty principle
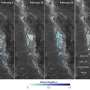
Anyone who has ever taken a group photo will be familiar with the problem: If everyone is constantly running around, it's almost impossible to get a sharp photo. Cell biologists who want to visualize molecular processes inside cells face a similar challenge. The molecules dance about at high speed. Receptors at the cell surface move within milliseconds, while vesicles transport proteins in seconds. Researchers at the Max Planck Institute of Molecular Physiology in Dortmund have now found a way to pinpoint the positions of individual molecules while at the same time measuring their activity and interactions in the same living cell. A dedicated cooling protocol on a microscope allows to pause cellular life at subzero temperatures, to let it continue to live again after warming. From the series of individual snapshots obtained, the researchers are able to form a precise spatial-temporal picture of the activity patterns of individual molecules within individual cells.
Fluorescence microscopy allows seeing where biological molecules are in cells. However, what Werner Heisenberg formulated for quantum physics to a certain extent has its analogy in biology: In the living state one can observe the collective movement of molecules in cells, which makes it however difficult to determine their exact positions. Paradoxically, the molecular dynamics that sustain life have to be halted to record the position of molecules using high-resolution fluorescence microscopy.
Living matter maintains its structure by energy consumption, which results in dynamic molecular patterns in cells that are difficult to observe by fluorescence microscopy, because the molecules are too numerous and their movements too fast. To tackle this problem a choice needs to be made: to precisely record the position of the molecules in a 'dead' state or to follow their collective behaviour in the living state. Although researchers have been able to stop movements in cells by chemical fixation, such methods lead to irreversible cell death and the acquired images of molecular patterns are not representative of a living system.
Gentle cooling and warming
Scientists led by Philippe Bastiaens of the Max Planck Institute Dortmund have now developed a method that allows them to observe nanometer-sized patterns of biomolecules such as proteins in an arrested but living state. To do so, they lower the temperature of living cells to completely suspend the macromolecular motion in cells. This state, called cryo-arrest, leaves sufficient time to capture high-resolution images of molecular patterns. By adding and removing a cryo-protectant during the cooling and warming processes, the scientists are able to reanimate the arrested cells. The biological processes then resume, and the cells live on.
The researchers are able to cryo-arrest cells several times in sequence and obtain multiple snapshots to reveal motion patterns. "Life is always on the move to maintain its shape and therefore impossible to record accurately. Nevertheless, it is possible to do so with our method of reversible cryo-arrest. In a lose way, we've overridden the biological uncertainty principle," says Bastiaens, head of the Systemic Cell Biology Department.
Signal proteins assemble in clusters
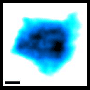
The researchers have been able to record not only the positions but also the activity and interactions of individual proteins. For example, they have found how EGF receptors – membrane proteins that transport growth factors inside cells and play an important role in carcinogenesis – often assemble in nanometer-sized groups on the cell surface. The scientists suspect that the receptors can be activated particularly easily in clusters so that they transmit a stronger growth signal into the interior of the cell.
In the next step, Bastiaens and his associates plan to refine the method and shorten the cooling process from several minutes to milliseconds in order to reversibly arrest cells without cryo-protectants.
More information: Reversible cryo-arrest for imaging molecules in living cells at high spatial resolution. Nature Methods (2016) DOI: 10.1038/nmeth.3921
Journal information: Nature Methods
Provided by Max Planck Society