May 27, 2016 report
Sequence-defined bioactive macrocycle can be tailored for drug design
Researchers from Cornell University have devised a peptidomimetic macrocyclic compound that is made from an acid-catalyzed cascade reaction. Their macrocycles are tunable in their backbone, side-chain composition and sequence, and their overall structure.
Using this structural versatility, Mintu Porel, Dana N. Thornlow, Ngoc N. Phan and Christopher A. Alabi designed a macrocycle that mimicked antimicrobial peptides and displayed antibacterial properties when tested with Gram-positive and Gram-negative bacteria. Their work has implications for making synthetic antibiotics. The report appears in Nature Chemistry.
According to Dr. Christopher A. Alabi, principle investigator of the study, "The emphasis of this work is sequence control with bioactive and biocompatible side chains that can be folded/tied at specific positions." He points out that the goal was not to recreate biological molecules but to capture their essential binding. For this study Dr. Alabi's group wanted to make compounds with bacterioloytic function the smallest sequence possible.
Macrocycles have unique properties that have proven effective in drug design. They are flexible ring structures that are comprised of at least twelve atoms and behave differently than other small molecules. For example macrocycles can penetrate the cell membrane even though they are often more than 500 Daltons in size. This is largely due to the flexible macrocycle's ability to fold into the right conformation to match the hydrophobicity of the surrounding environment.
Peptidomimetics are peptide-like polymers that are comprised of a backbone and individual, interchangeable monomers that are analogous to amino acids. In naturally-occurring peptides, the amino acid sequence determines how a protein will fold. Synthetic peptides, like the one used in the current study, can be designed such that a particular residue reacts with the tail end of the peptide sequence to make a closed ring.
In prior work Porel, et al. devised a synthesis for sequence defined oligothioetheramides (oligoTEAs) that relies on the addition of N-allylacryamide monomers to dithiols attached to a liquid alkyl fluoride (C9F19) support. In this work, they demonstrate the ability to make macrocyclic oligoTEAs using an acid-catalyzed (TFA) cascade reaction. The acid removes protecting groups from the alkoxyamine on the fluorinated terminal end as well as from a protected aldehyde monomer. The alkoxyamine reacts with the deprotected monomer, a reactive aldehyde, to form an oxime bond that closes the macrocyclic ring. Importantly, the protected monomer can be placed at any location along the polymer chain, allowing for macrocyclic rings of varying sizes with polymer tails of varying lengths.
Proton NMR and 2D 1H exchange spectroscopy demonstrated that the oligoTEA macrocycles had at least two different conformers. As the size of the ring decreased, the conformer exchange rate decreased indicating that the larger rings were more flexible, an important quality for synthetic drug design. The tail, when tail length reached a certain length, could also form cis or trans isomers.
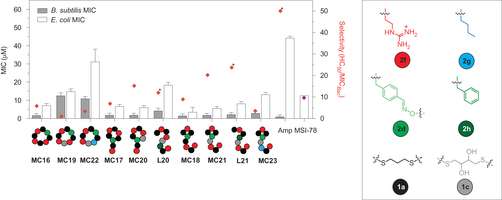
Porel, et al. demonstrated the synthetic versatility of their cascade reaction by making more than twenty different oligoTEA macrocycles. The N-allylacryamide monomer could tolerate various side chains including ones that allowed for tailoring the hydrophobicity and solubility of the macrocycle. The dithiol co-monomer could have two, three, or four carbons with hydroxyl groups. The monomer side chains, both the macrocycle and the tail, could be tailored for solubility and biological activity.
The next step was to conduct preliminary tests for antibacterial activity. Porel, et al. designed their oligoTEA to mimic antimicrobial peptides (AMPs) that are known to selectively penetrate the bacterial cell membrane. AMPs tend to have a hydrophobic and a hydrophilic face. Since the macrocyclic oligoTEA hydrophobicity can be tailored, this provides an excellent test for whether it can mimic AMP behavior.
Porel, et al. made eight new guanidinium-based oligoTEA macrocycles and tested their activity with Gram-negative (Escherichia coli) and Gram-positive (Bacillus subtilis) bacteria. All eight proved to be biostable to oxidation. They tended to be selective for Gram-positive bacteria, similar to other synthetic AMPs with smaller rings showing greater antibiotic activity than larger rings. Hemolysis studies indicated that two of the eight macrocycle oligoTEAs were selective for bacteria cells over red blood cells.
This research adds to the repertoire of synthetic drug design. Macrocyclic compounds have been a compelling area of research because their structure gives the molecules unique features that are conducive to crossing the cell membrane. Synthetic peptide-like polymers allow for tailoring the chemical properties and conformation of the macrocycle. The macrocyclic oligoTEAs presented here are made using a straight-forward synthesis that is versatile and relatively inexpensive. This research may provide an avenue for making new antibiotics.
More information: Mintu Porel et al, Sequence-defined bioactive macrocycles via an acid-catalysed cascade reaction, Nature Chemistry (2016). DOI: 10.1038/NCHEM.2508
Journal information: Nature Chemistry
© 2016 Phys.org