Parasites' winner-takes-all mechanism to evade immune defenses
A genetic game of 'winner takes all' that sustains the parasitic infections which cause sleeping sickness in humans and nagana in cattle – devastating diseases particularly in rural areas of Africa - has been identified in a research breakthrough led by the University of Dundee.
African trypanosomiasis, or sleeping sickness, affects both humans and animals and has a destructive impact, particularly in rural areas. The disease in humans is typically lethal without therapy and in cattle is the most economically important livestock disease in Africa.
The disease parasite is usually transmitted through the bite of an infected tsetse fly. At present, there is no vaccine. This is in part because the parasite cells – trypanosomal pathogens – are masters at evading the immune system.
"There's a constant 'arms race' between our immune system and pathogens, which involves constant change and adaptation on both sides," said David Horn, Professor of Parasite Molecular Biology in the School of Life Sciences at Dundee.
"Our immune system is generally very good at identifying and eliminating pathogens. But some parasites are stealthy and regularly switch what the immune system 'sees'. The parasites that cause trypanosomiasis and malaria are classic examples."
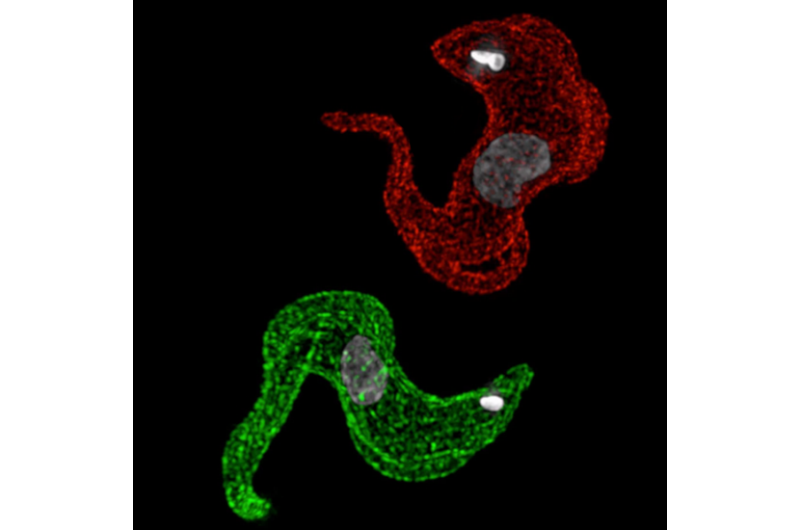
The African trypanosomes constantly undergo variation by changing their surface `protein coat' to escape immune system defenses.
They do this by sequentially expressing a family of variant genes encoding the different protein coats. The genes involved are among the largest families in parasite genomes, numbering into the thousands.
Professor Horn's team discovered a protein, which they call VEX1, that coordinates this expression of one type of protein coat at a time. VEX1 actually allows genes to compete with each other and helps just one to win the competition- a genetic version of 'winner takes all'.
"The various mechanisms allowing single gene choice in parasites and in our own cells have remained enigmatic," said Professor Horn.
"Having identified the trypanosome protein, we can now dissect the mechanism operating in these deadly parasites. We suspect that the mechanism will also share parallels with similarly enigmatic systems operating in other cells."
More information: Lucy Glover et al. VEX1 controls the allelic exclusion required for antigenic variation in trypanosomes, Proceedings of the National Academy of Sciences (2016). DOI: 10.1073/pnas.1600344113
Journal information: Proceedings of the National Academy of Sciences
Provided by University of Dundee