March 23, 2016 report
Study questions life extension mechanism in roundworms
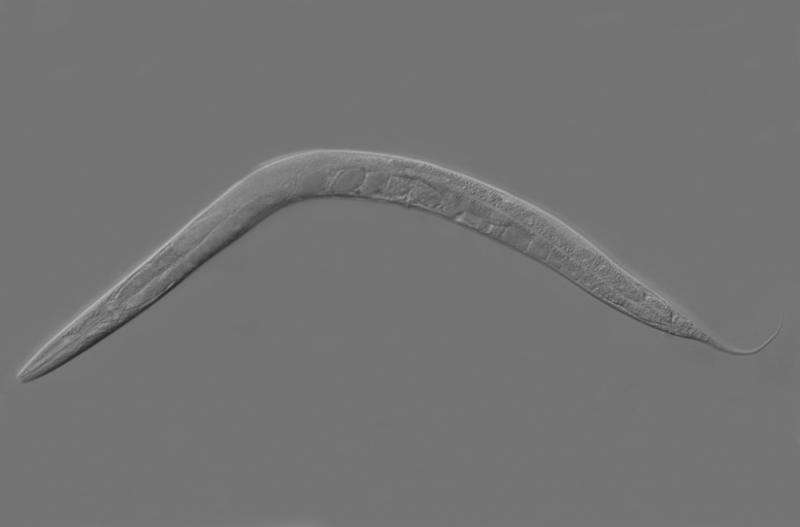
(Phys.org)—It's strange to think that there was a time when molecular cell biology was considered by professionals of other scientific specialties as disreputable or laughable. But the motivation for much cellular biology research around the mid-20th century was the search for the mechanisms of aging and the means to alter them—for centuries, people have been understandably frustrated by the indignities of aging and the burden of mortality, and the search for a "fountain of youth" was universally regarded as a foolish and hopeless endeavor.
It was only across decades of slow progress that the field was established as a fundamental source of health and medical science. (Watson and Crick certainly gave the field a boost with the discovery of DNA in 1953.) Now the discipline has exerted a sweeping and transformative effect across multiple fields including agriculture, food production and safety, pharmaceutical science, toxicology, human fertility, and biotechnology.
The mechanisms of cellular aging and longevity are slowly clarifying, and one of the avenues of interest is the distinction between germ cells and somatic cells in multicellular organisms. Germ cells give rise to gametes in sexually reproductive organisms. They are the only kind of cells that divide by meiosis as well as mitosis and are often called "immortal" because they are capable of generating entire new organisms across generations, while normal somatic cells differentiate into other cell types and are mortal, senescing and dying each generation.
Some researchers hope to capture germ cell programming and apply it to the somatic cells to prolong lifespan or even prevent death at the cellular level, and scientists seek examples of this dynamic in existing life forms. A study of daf-2 mutant Caenorhabditis elegans worms in 2009 suggested that they experience longer lifespans due to germ cell factors in their somatic cells. However, a new study questioning that result has found exactly the opposite: that expression of certain germ-line factors in the worms' somatic cells is actually detrimental to their health and results in a reduction of lifespan.
In the current study, published in the Proceedings of the National Academy of Sciences, researchers expressed puzzlement at the findings of the 2009 study. They write, "In our reevaluation of the Curran et al. findings, we took precautions to control for genetic background effects, which have been shown to influence lifespan, and we addressed key questions using multiple approaches... We found that daf-2 mutants do not detectably express germ-line proteins or transcripts in their somatic cells." They also note that lifespan analyses revealed that the worms don't use P granules or a particular master germ-line chromatin regulator to extend their lifespans.
The paper draws the obvious conclusion that the factors and molecular mechanisms responsible for germ-line immortality are not yet clearly understood. They write, "Future research will likely reveal how germ-line proteins influence these unique characteristics at the transcriptome and proteome levels. As the functions of various germ-line factors become clearer, a better understanding of the effects of misexpressing those factors in the soma will become clearer, as well."
More information: Reevaluation of whether a soma–to–germ-line transformation extends lifespan in Caenorhabditis elegans. PNAS 2016. DOI: 10.1073/pnas.1523402113
Abstract
The germ lineage is considered to be immortal. In the quest to extend lifespan, a possible strategy is to drive germ-line traits in somatic cells, to try to confer some of the germ lineage's immortality on the somatic body. Notably, a study in Caenorhabditis elegans suggested that expression of germ-line genes in the somatic cells of long-lived daf-2 mutants confers some of daf-2's long lifespan. Specifically, mRNAs encoding components of C. elegans germ granules (P granules) were up-regulated in daf-2 mutant worms, and knockdown of individual P-granule and other germ-line genes in daf-2 young adults modestly reduced their lifespan. We investigated the contribution of a germ-line program to daf-2's long lifespan and also tested whether other mutants known to express germ-line genes in their somatic cells are long-lived. Our key findings are as follows. (i) We could not detect P-granule proteins in the somatic cells of daf-2 mutants by immunostaining or by expression of a P-granule transgene. (ii) Whole-genome transcript profiling of animals lacking a germ line revealed that germ-line transcripts are not up-regulated in the soma of daf-2 worms compared with the soma of control worms. (iii) Simultaneous removal of multiple P-granule proteins or the entire germ-line program from daf-2 worms did not reduce their lifespan. (iv) Several mutants that robustly express a broad spectrum of germ-line genes in their somatic cells are not long-lived. Together, our findings argue against the hypothesis that acquisition of a germ-cell program in somatic cells increases lifespan and contributes to daf-2's long lifespan.
Journal information: Proceedings of the National Academy of Sciences
© 2016 Phys.org