'Disruptive device' brings xenon-NMR to fragile materials
Here's a new technology that's potentially disruptive precisely because it's non-disruptive: Scientists at the Department of Energy's Lawrence Berkeley National Laboratory (Berkeley Lab) have developed a device that enables NMR (nuclear magnetic resonance) spectroscopy, coupled with a powerful molecular sensor, to analyze molecular interactions in viscous solutions and fragile materials such as liquid crystals.
In a first, their method allows the sensor, hyperpolarized xenon gas, to be dissolved into minute samples of substances without disrupting their molecular order.
The technique brings the analytic power of hyperpolarized-gas NMR to materials that are too fragile to accept xenon gas through bubbling or shaking, which are the conventional delivery methods. It could help scientists learn more about advanced polymers, filters and catalysts for industrial processes, and liquid-crystal displays, to name a few applications.
The research was performed in the lab of NMR pioneer Alexander Pines, a senior faculty scientist with Berkeley Lab's Materials Sciences Division and UC Berkeley's Glenn T. Seaborg Professor of Chemistry. Ashley Truxal and Clancy Slack, who are UC Berkeley graduate students and members of Berkeley Lab's Materials Sciences Division, conducted the research with several other scientists.
Their work was published online March 8 in the journal Angewandte Chemie.
"Our device provides a new, robust way of introducing hyperpolarized xenon gas into a sample without perturbing the order of its molecules," says Pines. "It will allow us to use NMR to study new types of viscous and fragile materials, as well as materials that hierarchically aggregate into more complex structures, such as synthetic membranes and biological cells."

NMR spectroscopy, like its imaging cousin magnetic resonance imaging (MRI), uses superconducting magnets to polarize the alignment of the nuclear spins in a sample. When a radio frequency pulse is applied, the spins of the nuclei flip and then relax back to alignment, which produces a characteristic frequency of their own. The frequency is converted by NMR detectors into a spectral readout of the type, distribution, and reaction state of the molecules in the material.
Often, however, only a small percentage of the nuclear spins in a sample are polarized, which significantly limits NMR's sensitivity. One way to boost the strength and sensitivity of NMR signals is to hyperpolarize the nuclear spins, meaning the nuclei are polarized far beyond their thermal equilibrium conditions. The isotope xenon-129 is relatively easy to hyperpolarize and gives a large NMR signal in response to small changes in its surroundings. It's bubbled into a material that scientists want to analyze with NMR, and the spin of the xenon nuclei reports back what's inside.
But hyperpolarized xenon gas has one big limitation: when it's bubbled into a viscous solution or molecularly aligned material, the bubbles disrupt the sample, sometimes to the point of destroying it.
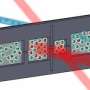
Berkeley Lab scientists have now overcome this limitation. Their method dissolves hyperpolarized xenon gas into fragile samples without wreaking havoc on their molecular order. Here's how it works: a sample to be studied is placed inside hollow silicone membrane fibers. Xenon gas is diffused through the columns, and only the xenon inside the columns is analyzed. The NMR signal is acquired, and the xenon gas diffuses out of the columns, to be replaced by new gas.
"Our system essentially breathes xenon in and out of the columns, so the signal source is constantly replenishing," says Truxal. "In addition to being non-disruptive to the sample, the approach requires a very small amount of sample, so the NMR analysis is very efficient."
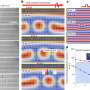
The scientists have demonstrated their non-disruptive approach on two materials that can't be probed by hyperpolarized xenon gas using conventional techniques. In one example, they used the device to track phase changes in MBBA, an organic liquid crystal.
"Understanding precisely when and why a liquid crystal undergoes a phase change can help us take advantage of the properties, perhaps leading to better electronic displays for example," says Truxal.
The scientists also used the device to analyze a bacteriophage with liquid crystalline properties, indicating the technique can be applied to a wide range of biological materials.
More information: Ashley E. Truxal et al. Nondisruptive Dissolution of Hyperpolarized Xe into Viscous Aqueous and Organic Liquid Crystalline Environments , Angewandte Chemie International Edition (2016). DOI: 10.1002/anie.201511539
Journal information: Angewandte Chemie International Edition
Provided by Lawrence Berkeley National Laboratory