Research sheds new light on structure of gold nanoparticles in water
Researchers at the University of Jyväskylä, Finland, and Colorado State University, USA, have for the first time ever determined the dynamical behaviour of the ligand layer of a water-soluble gold nanocluster in solution. The breakthrough opens a way towards controllable strategies for the functionalisation of ligated nanoparticles for applications. The work at the University of Jyväskylä was supported by the Academy of Finland. The research was published in Nature Communications on 21 January 2016.
Nanometre-scale gold particles are intensively investigated for applications as catalysts, sensors, drug delivery devices and biological contrast agents and as components in photonics and molecular electronics. The smallest particles have metal cores of only 1–2 nm with a few tens to a couple of hundred gold atoms. Their metal cores are covered by a stabilising organic ligand layer. The molecular formulas and solid-state atomic structure of many of these compounds, called "clusters", have been resolved during the past few years. Still, it is a considerable challenge to understand their atomic-scale structure and dynamical behaviour in the solution phase. This is crucial information that can help researchers understand how nanoclusters interact with the environment.
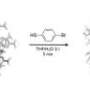
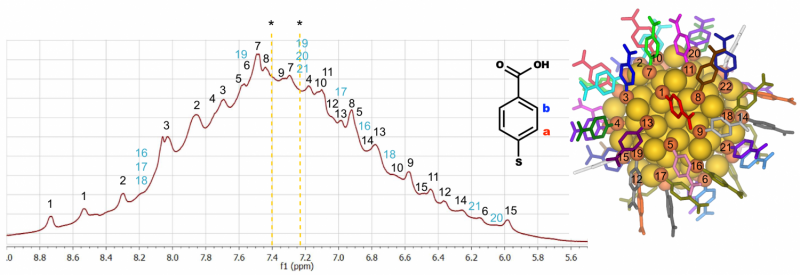
The researchers studied a previously identified molecularly precise nanocluster that has 102 gold atoms and 44 thiol ligands (Figure 1, right). The solid-state structure of this cluster was resolved from single-crystal X-ray diffraction experiments in 2007. The ligand shell has a low symmetry and produces a large number of signals in conventional proton-NMR measurement (Figure 1, left). The researchers achieved a full assignment of all signals to specific thiol ligands by using a combination of correlated nuclear magnetic resonance (NMR) experiments, density functional theory computations and molecular dynamics simulations.
The Finnish researchers at Jyväskylä have previously used this specific cluster material, for instance, for structural studies of enteroviruses.
"Now that we know exactly which ligand produces which NMR signal, we can proceed with precise studies on how this nanocluster interacts with the chemical and biological environment in the water phase. This gives unprecedented potential to understand and control the inorganic-organic interfaces that are relevant to hybrid inorganic-biological materials," says Academy Professor Hannu Häkkinen from the Nanoscience Center at the University of Jyväskylä. Häkkinen coordinated the work of the Finnish-US team.
More information: Kirsi Salorinne et al. Conformation and dynamics of the ligand shell of a water-soluble Au102 nanoparticle, Nature Communications (2016). DOI: 10.1038/ncomms10401
Journal information: Nature Communications
Provided by Academy of Finland