December 11, 2015 feature
Study shows important goal for organic semiconductors is attainable

(Phys.org)—In organic semiconductor materials, there has always been a tradeoff between the ability to emit light and the ability to conduct a charge, as measured by the charge carrier mobility. Now for the first time, scientists from China and the UK have designed and synthesized a new type of organic semiconductor that combines both high luminescence and high mobility in a single material.
The researchers, Jie Liu, et al, have published a paper on the new organic semiconductor in a recent issue of Nature Communications.
"The invention of an organic semiconductor with high mobility and strong emission opens the doors to the design and synthesis of [additional] novel organic semiconductors," coauthor Huanli Dong at the Chinese Academy of Sciences and Beihang University, both in Beijing, told Phys.org.
Currently, all organic semiconductor materials with high luminescence exhibit low mobility, and vice versa. Combining both traits in a single organic semiconductor has been an important goal because the combination is essential for developing new types of optoelectronics devices, such as organic light-emitting transistors (OLETs) and their offshoots, including OLET-based displays and organic electrical pumping lasers based on OLETs.
The reason why high mobility and high luminescence don't mix is because they generally require opposite types of molecular structures. High mobility requires that molecules pack together densely, but densely packed molecules cause "fluorescence quenching." Fluorescence occurs when an electron falls to its ground state, emitting a photon in the process. However, densely packed molecules tend to have strong intermolecular interactions that prevent electrons from transitioning to their ground states, which prevents photon emission.
When designing their new organic semiconducting compound, called 2,6-diphenylanthracene (DPA), the researchers kept the molecules closely packed to ensure a high mobility. But by making the molecules pack together in a strategic way, the researchers could also greatly reduce the fluorescence quenching.
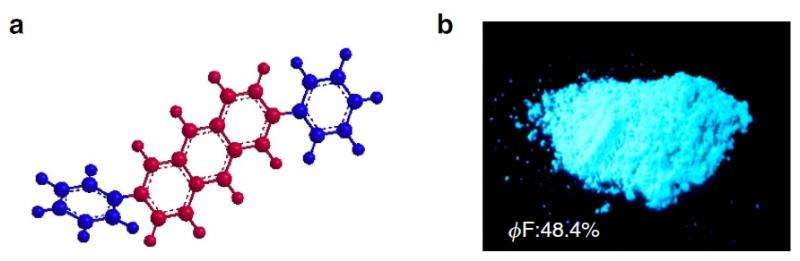
The researchers describe the new molecular arrangement as "herringbone packing," a zig-zag pattern that has weaker intermolecular interactions than other types of arrangements, even though the molecules are still very close together. The weaker interactions allow electrons to transition to the ground state, so the molecule exhibits a high luminescence along with a high mobility.
To demonstrate these traits, the researchers used the DPA semiconductor to fabricate organic light-emitting diodes (OLEDs) that highlight DPA's bright blue luminescence, as well as organic field-effect transistors (OFETs) that show DPA's good charge transport. They also integrated these two devices, creating an OFET-driven OLED array based on the same organic semiconductor, DPA, to demonstrate the potential of using the new material in organic optoelectronics devices.
More information: Jie Liu, et al. "High mobility emissive organic semiconductor." Nature Communications. DOI: 10.1038/ncomms10032
Journal information: Nature Communications
© 2015 Phys.org