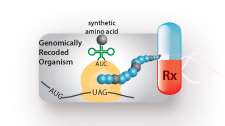
Using bacterial genomes that have been entirely recoded, Yale scientists have developed a way to induce cells to produce novel protein-based polymers that can be used for new materials and therapeutics.
Cells usually assemble proteins using 20 amino acids and scientists have found it difficult to incorporate synthetic amino acids in more than a single or few positions in proteins. The new technology, described Nov. 16 in the journal Nature Biotechnology, shows how it is possible to incorporate many new synthetic amino acids into proteins and thus go beyond the standard 20 building blocks to endow completely new function.
No longer will scientists be limited in the quantity and diversity of novel amino acids they can use to make new therapeutics and materials in bacterial factories, the authors say.
"Now we can introduce dozens of new synthetic building blocks with user-defined precision and at will—as many times as we want—to produce new compounds to improve drugs or functionalized polymers for new materials, for example biocompatible glue," said Farren Isaacs, senior author and assistant professor of molecular, cellular and developmental biology at the Systems Biology Institute at Yale's West Campus.
In the last few years, scientists at Yale and elsewhere have successfully recoded bacterial organisms to improve incorporation of synthetic amino acids in proteins. Isaacs, lead author Miriam Amiram, co-senior authors Jesse Rinehart and Dieter Söll of Yale, and the team of researchers from Yale and Northwestern University designed new cellular machinery that can encode synthetic amino acids with high efficiency and of high purity into proteins.
More information: Miriam Amiram et al. Evolution of translation machinery in recoded bacteria enables multi-site incorporation of nonstandard amino acids, Nature Biotechnology (2015). DOI: 10.1038/nbt.3372
Journal information: Nature Biotechnology
Provided by Yale University