Biologists report method to calculate lifetime energy requirements of cells, genes
In a recently published paper, Indiana University biologists have calculated the lifetime energy requirements of multiple types of cells, as well as the energy required to replicate and express the genes within these cells.
"The Bioenergetic Costs of a Gene," reported in the Proceedings of the National Academy of Sciences, describes for the first time how much total energy is needed to build and maintain a cell, and how this scales with cell size.
"This study attempts to systematically map out the lifetime energy requirements of cells in a comprehensive way across a multitude of species, as well as the total cost of maintaining and expressing a genome," said IU biologist Michael Lynch, who led the study. "It also negates in a solid way a common argument that complex cells could never have evolved without an energy-producing mitochondrion."
Lynch is a Distinguished Professor in the IU Bloomington College of Arts and Sciences' Department of Biology. Georgi K. Marinov, a postdoctoral researcher in biology, is co-author on the paper.
The research is part of a larger effort in Lynch's lab to calculate the precise energy cost of every part of a cell, including the cellular membrane and internal organelles.
The first step, reported in the paper, describes the total energy costs of individual genes, as incurred at the DNA, RNA and protein levels.
To conduct the study, Lynch and Marinov reviewed papers going back as far as the 1960s, each focusing on different organisms, and applied current information on metabolic pathways and single-cell attributes to figure out each cell's lifetime energy needs. They mapped out both "the energy required to maintain the cell" and "the energy required to build all the parts of the next offspring cell," Lynch said. The goal was to "find the denominator," or the total amount of energy by which every other structure's needs would be divided.
The unit of energy used in the study was ATP, or adenosine nucleoside triphosphate, a simple molecule that serves as the major carrier of cellular energy in cells through the breakage of phosphate bonds. In terms of energy, a single ATP molecule produces about ten quintillionth of a joule when one of these bonds is broken.
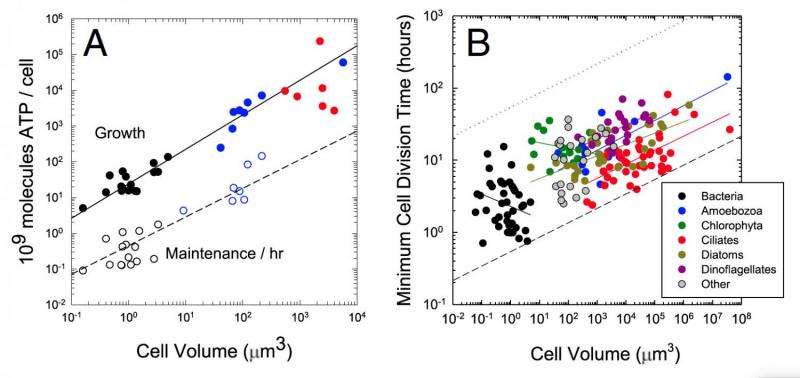
The simplest cells in the study, bacteria, needed about 100 million molecules of ATP energy over the course of their life. The most complex cells in the study, eukaryotic ciliates, needed more than 1,000 times that amount across their lifespan.
In eukaryotes, ATP is mostly produced by the mitochondrion, which is the cellular structure responsible for the majority of energy utilization in complex eukaryotic cells.
Because they possess a genome independent of their host cells, mitochondria are known to have started out as an ancient bacteria absorbed into early single-celled organisms about 2 billion years ago.
Symbiogenesis is the term used to describe the concept that cells without nuclei or organelles—prokaryotes—gave rise to more complex cells with nuclei and organelles—eukaryotes—after a single-celled organism absorbed a bacterial ancestor of the mitochondrion. It is regarded as one of the most important events in evolutionary history.
Surprisingly, however, the IU scientists found that energy consumption grew linearly with cell volume, not exponentially or stepwise, as cells grew more complex. So while eukaryotic cells require more energy for cellular reproduction, they didn't increase their energy needs at a higher rate relative to cell volume compared to prokaryotic cells.
"The enhanced ability to generate energy made possible by the mitochondrion is often seen as a prerequisite for the evolution of eukaryotic cell features such as increased gene number, protein length, protein folds, protein-protein interactions and regulatory elements," Lynch said. "But our observations suggest that the energy boost associated with the mitochondrion is not a precondition for genome complexity."
The ultimate goal of the researchers' project is similar to those of a homeowner who wants to know the cost of building a house, Lynch said.
"If you've got a limited budget, you can't start construction until you know the costs of all the materials," Lynch said. Similarly, evolution isn't free, and building every part of a cell requires costs that could be used elsewhere.
Understanding how these costs versus benefits parcel out will help shed light on how forces such as natural selection might have influenced the vast cellular diversity found on Earth.
If the costs of building a structure are about the same as the benefits acquired, for example, then there is no net gain, or loss, of fitness in the long run.
"This work links to a substantial body of theory outlining increasingly plausible scenarios by which complex traits may emerge by mechanisms other than natural selection," Lynch said. "We can no longer simply assume that all features of organisms arise from promotion by natural selection."
More information: Michael Lynch et al. The bioenergetic costs of a gene, Proceedings of the National Academy of Sciences (2015). DOI: 10.1073/pnas.1514974112
Journal information: Proceedings of the National Academy of Sciences
Provided by Indiana University