Chipping away at the secrets of ice formation
Making ice to chill our drinks is easy enough, but surprisingly, the details of that seemingly simple process are still not well understood. Now scientists report in the Journal of the American Chemical Society that they have unlocked some of ice's mysteries while debunking a major assumption about the process. The findings could help us better understand how water freezes, which has significant implications for climate science, air travel and cryopreservation.
When it comes to water for drinking and cooking, cleaner is better. But when it comes to cloud formation and rain and snow, impurities are a big help. Tiny particles such as dust, soot, pollen and even bacteria give water a seed to crystalize around. Without them, water can remain liquid even at temperatures below its freezing point. So what kinds of particles work best to promote ice formation? Angelos Michaelides and colleagues searched for an answer.
Using computer models of simple crystalline particle surfaces, the researchers pinpointed how their structures and their interactions with water on a molecular level impact ice crystallization. A commonly held assumption was that if a particle's surface structure is similar to that of ice, it acts as a template for water molecules and encourages ice to form. But the team found that a surface that acts as a template for one ice face will not necessarily act as a template for another. And two identical surfaces can result in ice crystals in three different orientations. In addition to informing the more obvious fields of climate science and cryopreservation, the researchers say their results could help guide the future design of materials to prevent ice formation, for example, on airplane wings.
More information: Martin Fitzner et al. The Many Faces of Heterogeneous Ice Nucleation: Interplay Between Surface Morphology and Hydrophobicity, Journal of the American Chemical Society (2015). DOI: 10.1021/jacs.5b08748
Abstract
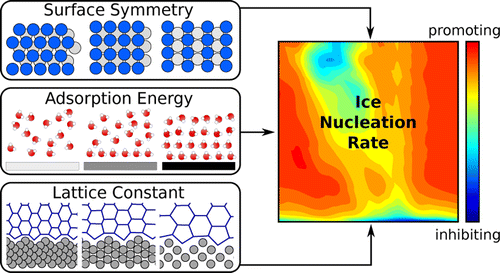
What makes a material a good ice nucleating agent? Despite the importance of heterogeneous ice nucleation to a variety of fields, from cloud science to microbiology, major gaps in our understanding of this ubiquitous process still prevent us from answering this question. In this work, we have examined the ability of generic crystalline substrates to promote ice nucleation as a function of the hydrophobicity and the morphology of the surface. Nucleation rates have been obtained by brute-force molecular dynamics simulations of coarse-grained water on top of different surfaces of a model fcc crystal, varying the water–surface interaction and the surface lattice parameter. It turns out that the lattice mismatch of the surface with respect to ice, customarily regarded as the most important requirement for a good ice nucleating agent, is at most desirable but not a requirement. On the other hand, the balance between the morphology of the surface and its hydrophobicity can significantly alter the ice nucleation rate and can also lead to the formation of up to three different faces of ice on the same substrate. We have pinpointed three circumstances where heterogeneous ice nucleation can be promoted by the crystalline surface: (i) the formation of a water overlayer that acts as an in-plane template; (ii) the emergence of a contact layer buckled in an ice-like manner; and (iii) nucleation on compact surfaces with very high interaction strength. We hope that this extensive systematic study will foster future experimental work aimed at testing the physiochemical understanding presented herein.
Journal information: Journal of the American Chemical Society
Provided by American Chemical Society