Bend me, shape me, any way you want me: Scientists curve nanoparticle sheets into complex forms

Scientists have been making nanoparticles for more than two decades in two-dimensional sheets, three-dimensional crystals and random clusters. But they have never been able to get a sheet of nanoparticles to curve or fold into a complex three-dimensional structure. Now researchers from the University of Chicago, the University of Missouri and the U.S. Department of Energy's Argonne National Laboratory have found a simple way to do exactly that.
The findings open the way for scientists to design membranes with tunable electrical, magnetic and mechanical properties that could be used in electronics and may even have implications for understanding biological systems.
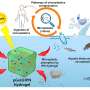
Working at the Center for Nanoscale Materials (CNM) and the Advanced Photon Source (APS), two DOE Office of Science User Facilities located at Argonne, the team got membranes of gold nanoparticles coated with organic molecules to curl into tubes when hit with an electron beam. Equally importantly, they have discovered how and why it happens.
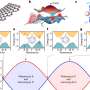
The scientists coat gold nanoparticles of a few thousand atoms each with an oil-like organic molecule that holds the gold particles together. When floated on water the particles form a sheet; when the water evaporates, it leaves the sheet suspended over a hole. "It's almost like a drumhead," says Xiao-Min Lin, the staff scientist at the Center for Nanoscale Materials who led the project. "But it's a very thin membrane made of a single layer of nanoparticles."
To their surprise, when the scientists put the membrane into the beam of a scanning electron microscope, it folded. It folded every time, and always in the same direction.
"That got our curiosity up," said Lin. "Why is it bending in one direction?"
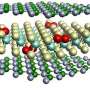
The answer lay in the organic surface molecules. They are hydrophobic: when floated on water they try to avoid contact with it, so they end up distributing themselves in a non-uniform way across the top and bottom layers of the nanoparticle sheet. When the electron beam hits the molecules on the surface it causes them to form an additional bond with their neighbors, creating an asymmetrical stress that makes the membranes fold.
Zhang Jiang and Jin Wang, X-ray staff at the APS, came up with an ingenious way to measure the molecular asymmetry, which at only six angstroms, or about six atoms thick, is so tiny it would not normally be measurable.
Subramanian Sankaranarayanan and Sanket Deshmukh at CNM used the high-performance computing resources at DOE's National Energy Research Scientific Computing Center and the Argonne Leadership Computing Facility (ALCF), both DOE Office of Science User Facilities, to analyze the surface of the nanoparticles. They discovered that the amount of surface covered by the organic molecules and the molecules' mobility on the surface both have an important influence on the degree of asymmetry in the membrane.
"These are fascinating results," said Fernando Bresme, professor of chemical physics at the Imperial College in London and a leading theorist on soft matter physics. "They advance significantly our ability to make new nano-structures with controlled shapes."
In principle, scientists could use this method to induce folding in any nanoparticle membrane that has an asymmetrical distribution of surface molecules. Said Lin, "You use one type of molecule that hates water and rely on the water surfaces to drive the molecules to distribute non-uniformly, or you could use two different kinds of molecules. The key is that the molecules have to distribute non-uniformly."
The next step for Lin and his colleagues is to explore how they can control the molecular distribution on the surface and therefore the folding behavior. They envision zapping only a small part of the structure with the electron beam, designing the stresses to achieve particular bending patterns.
"You can maybe fold these things into origami structures and all sorts of interesting geometries," Lin said. "It opens the possibilities."
Provided by Argonne National Laboratory