July 27, 2015 report
Researchers find short tracks of DNA may aid in regulating human gene expression
(Phys.org)—A team of researchers with members from Washington University, Johns Hopkins School of Medicine, the Howard Hughes Medical Institute and the Polish Academy of Sciences has found that problems with RNA appear to be behind protein translation interruptions and that short segments of DNA may assist in regulating gene expression. They have published a paper describing their research and findings in the journal Science Advances.
Scientists have known for some time that the mechanism that controls protein translation, known as polyadenylate A aka, poly(A) is sometimes interrupted, causing degradation of messenger RNA (mRNA) and the proteins under development, leading to some ailments such as neurodegenerative diseases. Past research has suggested the problem lies with amino acids involved in the encoding, but now it appears that the problem is actually with the RNA itself—specifically strings of multiple adenosine (A) nucleotides.
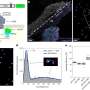
In this new effort the team, noting that approximately 2 percent of genes in the human genome may be impacted, found that in studying bacterial ribosomes, that they were more likely to be interrupted on strings of lysines if they were encoded by AAA codons, as opposed to AAG codons. They showed that making shorter or longer runs of adenosine nucleotides, without modifying amino acid sequences, changed the protein output and also the stability of the mRNA. They noted also that doing so also sometimes led to the creation of what they termed "frameshifted" protein products.
The researchers also studied poly(A) tracks in human cells and found some as short as just nine basses long might influence gene expression. They discovered that poly(A) lowered the expression of protein in two different ways. The first was by halting translation, which led to degradation of the protein and mRNA itself. The second was when frameshifts occurred during translation, which led to early termination of the production of proteins.
The work by the team, and another also at Washington University looking into the impact of nucleotides on Poly(A) offer a fresh insight into the creation of disease-causing states in cells and by extension, possible ways to prevent it from happening, offering patients with such ailments hope of recovery.
More information: Translational control by lysine-encoding A-rich sequences, Science Advances 24 Jul 2015: Vol. 1, no. 6, e1500154 DOI: 10.1126/sciadv.1500154
Abstract
Regulation of gene expression involves a wide array of cellular mechanisms that control the abundance of the RNA or protein products of that gene. We describe a gene regulatory mechanism that is based on polyadenylate [poly(A)] tracks that stall the translation apparatus. We show that creating longer or shorter runs of adenosine nucleotides, without changes in the amino acid sequence, alters the protein output and the stability of mRNA. Sometimes, these changes result in the production of an alternative "frameshifted" protein product. These observations are corroborated using reporter constructs and in the context of recombinant gene sequences. About 2% of genes in the human genome may be subject to this uncharacterized yet fundamental form of gene regulation. The potential pool of regulated genes encodes many proteins involved in nucleic acid binding. We hypothesize that the genes we identify are part of a large network whose expression is fine-tuned by poly(A) tracks, and we provide a mechanism through which synonymous mutations may influence gene expression in pathological states.
Journal information: Science Advances
© 2015 Phys.org