RNA-binding protein influences key mediator of cellular inflammation and stress responses
Messenger (mRNA) molecules are a key component of protein biosynthesis. They are first transcribed as a "working copy" of the DNA and then translated into protein molecules. RNA-binding proteins such as RC3H1 (also known as ROQUIN) regulate the degradation of the mRNA molecules and thus prevent the production of specific proteins. Researchers at the Max Delbrück Center for Molecular Medicine in the Helmholtz Association (MDC) have now shown that ROQUIN binds several thousand mRNA molecules. They demonstrated that ROQUIN also influences the gene regulator NF-kappaB, a key mediator of cellular inflammation and stress responses (Nature Communications, Article number: 7367).
RC3H1/ROQUIN has already been described in previous studies as an RNA-binding protein that influences the stability of various mRNAs. Until now, however, it was unclear how ROQUIN recognizes mRNAs and how many mRNAs are regulated by ROQUIN. Dr. Yasuhiro Murakawa and Dr. Markus Landthaler of the Berlin Institute for Medical Systems Biology (BIMSB) of the MDC, in collaboration with the MDC research groups led by Professor Udo Heinemann, Dr. Stefan Kempa, Professor Claus Scheidereit, Dr. Jana Wolf and others, showed that ROQUIN binds to more than 3,800 different mRNAs, and they identified more than 16,000 sites to which the protein binds. Thus, ROQUIN appears to have greater influence on the regulation of gene expression than previously thought. In addition, the researchers identified the RNA recognition sequences of ROQUIN, thus providing insight into where protein-RNA interactions take place.
ROQUIN regulates the response to DNA damage
ROQUIN preferentially binds mRNAs generated in response to DNA damage but also in the context of inflammatory responses. Many of the affected mRNAs encode for proteins, which in turn influence the activity of genes and thus regulate the production of other proteins. According to the researchers, ROQUIN contributes to the fine-tuning of the regulatory mechanisms.
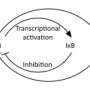
One of the target transcripts of ROQUIN is the mRNA coding for the protein A20 (also known as TNFAIP3). A20 serves as feedback control of the IkappaBalpha-kinase complex (IKK) that regulates the activation of the gene regulator NF-kappaB. The IKK/NF-kappaB pathway regulates the expression of a number of genes and is one of the key mediators in inflammatory and cellular stress responses, e.g. induced by DNA damage.
To prevent the sustained activation of the IKK/NF-kappaB pathway, this signaling pathway itself induces the increased expression of proteins such as A20, accompanied by decreased IKK/NF-kappaB activation. By regulating the decay of the mRNA for A20, ROQUIN thus indirectly modulates the activity of the IKK/NF-kappaB pathway.
Better understanding of autoimmune diseases
The researchers hypothesize that ROQUIN may play an even more extensive functional role and is involved in other signaling pathways, where it shortens the life span of protein-encoding mRNAs and thus enables fine-tuning. These findings are important to better understand and possibly prevent autoimmune diseases. The protein A20, for example, is known as a protective factor against arthritis. Knockdown of ROQUIN, which results in increased A20 protein expression, may therefore represent an approach to treating this chronic joint inflammation disease.
Journal information: Nature Communications
Provided by Helmholtz Association of German Research Centres