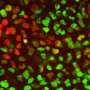
Toronto scientists have recorded, in unprecedented detail, the locations of all proteins in a cell. This new protein map allows scientists to look much more closely into what happens in a cell when disease strikes, and will also help find better treatments.
Led by Professors Brenda Andrews, Charles Boone, and Jason Moffat from the University of Toronto's Donnelly Centre, the team built a state-of-the-art automated pipeline to monitor where proteins sit in the cell and to see how they move in response to genetic or environmental perturbations.
The study is published in today's issue of Cell, a leading journal in the field.
The detailed database of protein locations will be made available, also this month, through G3: Genes|Genomes|Genetics, the official journal of the Genetics Society of America, so that anyone can look up location and movement of their protein(s) of interest.
As cells do their jobs, such as making, maintaining and repairing our bodies, they continuously move proteins around. But scientists understand very little about how this traffic occurs inside our cells. This is about to change as the new map, which charts protein movement and abundance, becomes available. Much as the shipping or airline routes give insights into the state of world economy, so this new protein map will help scientists understand better what happens in cells when they are healthy and what goes wrong in a disease.
Proteins are products of genes and they are responsible for all the workings of the cell.
Credit: Erin Howe
"A lot of the regulation that happens within cells, which is critical for the basic functioning of the human body, influences where individual proteins are localized and how they move around. It is very important to understand how this regulation happens if we are going to be able to understand why cells are healthy and why they are sometimes diseased," says Brenda Andrews, who is also a professor in U of T's Department of Molecular Genetics.
To visualize and count most of the roughly 6000 proteins in the cell, researchers collected data for mind-boggling 20 million cells. For more than a decade, the scientists worked closely together with robotic engineers, who built machines to handle the cells, and software writers who designed artificial intelligence-based algorithms to process the vast amount of data.
"The reason we need to do it on a large scale is because there simply are so many proteins," says Andrews, who uses baker's yeast as a model to understand human cell biology.
Yeast cells work in very similar ways to human cells but have fewer proteins, around a quarter the number that exist in more complex human cells. This relative simplicity has allowed researchers like Andrews and Boone to use yeast to make many fundamental insights into how both yeast and human cells work.
Their team not only charted protein movement and abundance in normal cells, but they also looked at what happen when cells carry a mutation, which could lead to a genetic disease, for example, or when they are exposed to different drugs.
"We've developed methods that allow scientists to examine all of proteins in the cell and how they change in response to any kind of perturbation," says Andrews.
Next, the researchers will use this powerful pipeline to investigate how proteins move in human cells, such as cancer cells, to understand better the origin of the disease, but also to search for new treatments.
"We want to understand how all proteins are moving, at a systems level, in cancer cells upon, say, a treatment with a drug or genetic perturbation, so that we can identify vulnerabilities in cancer cells, in terms of protein localization and abundance, and start thinking about how to best target those changes," says Moffat, also a professor in U of T's Department of Molecular Genetics.
Journal information: Cell
Provided by University of Toronto