Manipulating cell membranes using nanotubes
Japanese researchers have developed a targeted method for opening up cell membranes in order to deliver drugs to, or manipulate the genes of, individual cells.
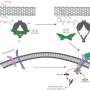
The method, as reported in the journal Science and Technology of Advanced Materials, involves irradiating a thin film of carbon nanotubes using a near-infrared (NIR) laser. The nanotubes act as an effective photon absorber as well as a stimulus generator for adjacent cells.
In cell engineering and tissue biology research, scientists often use pulsed lasers to stimulate cells and enable gene transfection (the introduction of genetic material), gene regulation or drug injection. The irradiation of biological cells using pulsed lasers causes their membranes to puncture, which significantly accelerates gene transfection or the targeted delivery of drugs.
Among the wide range of photon energies, the near-infrared region is less harmful for biological cells, which absorb very little energy in these wavelengths. The most successful NIR lasers are femtosecond lasers due to their fine spatial resolution with no thermal or mechanical damage to surrounding materials. However, femtosecond laser instruments are expensive, bulky and require a highly sophisticated optical arrangement, so the research team opted for a more economical nanosecond laser.
In the study, Naotoshi Nakashima and colleagues at Kyushu University prepared a dish coated with single-walled carbon nanotubes, which strongly absorb radiation in the NIR region, as an antenna for a nanosecond pulse laser. The dish was also seeded with living cells.
Depending on the energy of the laser, the researchers found that the cell membranes were either reversibly or irreversibly disturbed. When a laser pulse exceeded 17.5 microJoules (uJ), the membranes were destroyed and the cells died. However, at lower energies of about 15 uJ per pulse, the membranes opened and the cells remained alive. This suggests that an inexpensive laser source could be used to prepare a single cell target for selective gene transfection, drug injection or regulation of gene expression, the authors conclude.
Journal information: Science and Technology of Advanced Materials
Provided by Kyushu University