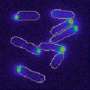
Small signaling molecule gives green light for cell division
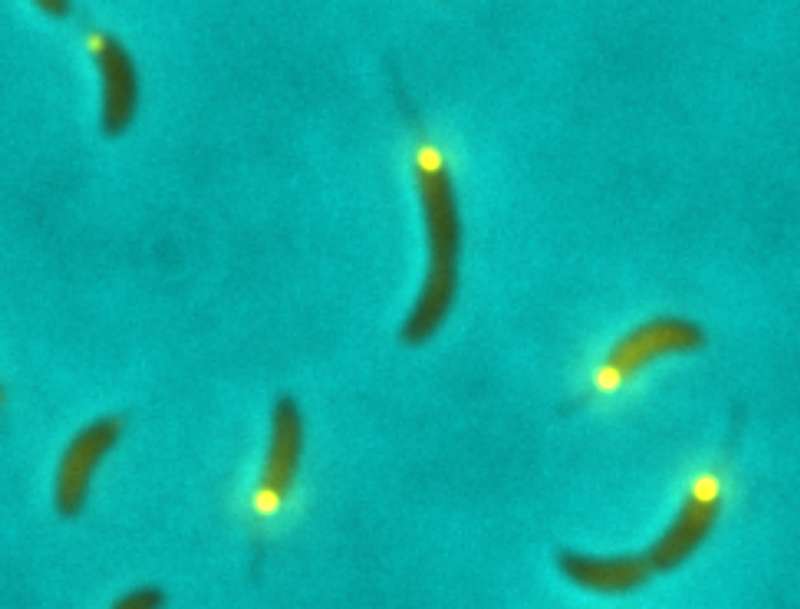
Generating offspring is the evolutionary goal of all living organisms. The multiplication of individual cells is coordinated by the cell cycle. For the discovery of how this process is regulated in eukaryotes the Noble Prize was awarded in 2001. The team of Prof. Urs Jenal at the Biozentrum of the University of Basel has now identified the central switch for reproduction in bacteria. While cell cycle progression in eukaryotes is regulated by small proteins called cyclins, in bacteria this role is adopted by a small signaling molecule, c-di-GMP. In the current issue of Nature the scientists describe the molecular details of this process.
Though very tiny, the molecule is vital for the survival of almost all bacteria. This signaling messenger – called c-di-GMP – controls behavioral processes in bacteria. For instance, it ensures that bacteria join together to form biofilms, which can cause chronic infections in humans. The scientists working with Prof. Urs Jenal at the Biozentrum of the University of Basel have now demonstrated that c-di-GMP also plays a decisive role in bacterial reproduction. They discovered that oscillating levels of the messenger subsequently influence the activity of key regulatory proteins, thereby controlling cell cycle progression and proliferation of bacteria.
Signaling molecule sets traffic lights at check points
How do cells multiply? When cells divide, two daughter cells arise from one mother cell. Before this, however, the cell must go through several phases from growth, to the replication of its genetic information and finally to cell division. This process is known as the cell cycle. In their study on the model bacterium Caulobacter crescentus the infection biologists show for the first time, that the signaling messenger c-di-GMP controls the cell cycle in a similar way as a traffic light works. In the absence of c-di-GMP in the cell, the light shows red. This indicates that the cell will have to remain in the first phase of the cell cycle. If the c-di-GMP level increases, the light switches to green and the cell enters the next phase. The scientists have investigated what exactly occurs on the molecular level.
c-di-GMP controls an enzyme with two modes of action
The role of this traffic light is played by an enzyme that works in two different ways. "When c-di-GMP is lacking, it blocks the process which leads to replication of the genetic material," explains Jenal. "However, as soon as c-di-GMP is produced, it binds to the enzyme, thus altering its structure and mode of action. Subsequently, this blockade is lifted and the bacterial chromosomes can be copied." This step marks the entry into the next phase of the cell cycle. The varying spatial distribution of the signaling molecule in the dividing mother cell also plays an important role in the behavior of the progeny.
Pathogens use the same signaling network
It is the first time that the researchers have been able to establish a direct connection between the two major regulatory networks of bacterial cells, – the small messenger and important regulatory enzymes called kinases. The insights gained provide an important basis for elucidating the much more complicated c-di-GMP networks of pathogens. The signaling molecule is involved in virulence, persistence mechanisms and antibiotic resistance of pathogens. For instance, dangerous pathogens causing cholera or pneumonia use c-di-GMP signaling to survive in their human host. As a next step, the researchers want to figure out, whether this molecule acts in these pathogens in the same way as in the model bacterium C. crescentus.
More information: "Cyclic di-GMP acts as a cell cycle oscillator to drive chromosome replication" Nature (2015) DOI: 10.1038/nature14473
Journal information: Nature
Provided by University of Basel