Researchers discover the secret to substances that can be engineered to 'freeze' at a specific time

Water, when cooled below 32°F, eventually freezes—it's science known even to pre-schoolers. But some substances, when they undergo a process called "rapid-freezing" or "supercooling," remain in liquid form—even at below-freezing temperatures.
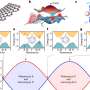
The supercooling phenomenon has been studied for its possible applications in a wide spectrum of fields. A new Tel Aviv University study published in Scientific Reports is the first to break down the rules governing the complex process of crystallization through rapid-cooling. According to the research, membranes can be engineered to crystallize at a specific time. In other words, it is indeed possible to control what was once considered a wild and unpredictable process—and it may revolutionize the delivery of drugs in the human body, providing a way to "freeze" the drugs at the exact time and biological location in the body necessary.
The study was led jointly by Dr. Roy Beck of the Department of Physics at TAU's School of Physics and Astronomy and Prof. Dan Peer of the Department of Cell Research and Immunology at TAU's Faculty of Life Sciences, and conducted by TAU graduate students Guy Jacoby, Keren Cohen, and Kobi Barkai.
Controlling a metastable process
"We describe a supercooled material as 'metastable,' meaning it is very sensitive to any external perturbation that may transform it back to its stable low-temperature state," Dr. Beck said. "We discovered in our study that it is possible to control the process and harness the advantages of the fluid/not-fluid transition to design a precise and effective nanoscale drug encapsulating system."
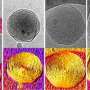
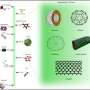
For the purpose of the study, the researchers conducted experiments on nanoscale drug vesicles (fluid-filled sacs that deliver drugs to their targets) to determine the precise dynamics of crystallization. The researchers used a state-of-the-art X-ray scattering system sensitive to nanoscale structures.
"One key challenge in designing new nano-vesicles for drug delivery is their stability," said Dr. Beck. "On the one hand, you need a stable vesicle that will entrap your drug until it reaches the specific diseased cell. But on the other, if the vesicle is too stable, the payload may not be released upon arrival at its target."
"Supercooled material is a suitable candidate since the transition between liquid and crystal states is very drastic and the liquid membrane explodes to rearrange as crystals. Therefore this new physical insight can be used to release entrapped drugs at the target and not elsewhere in the body's microenvironment. This is a novel mechanism for timely drug release."
All in the timing
The researchers found that the membranes were able to remain stable for tens of hours before collectively crystallizing at a predetermined time.
"What was amazing was our ability to reproduce the results over and over again without any complicated techniques," said Dr. Beck. "We showed that the delayed crystallization was not sensitive to minor imperfection or external perturbation. Moreover, we found multiple alternative ways to 'tweak the clock' and start the crystallization process."
The researchers are investigating an appropriate new nano-capsule capable of releasing medication at a specific time and place in the body. "The challenge now is to find the right drugs to exploit our insights for the medical benefit of patients," said Dr. Beck.
Journal information: Scientific Reports
Provided by Tel Aviv University