April 20, 2015 report
Plausibility of the vibrational theory of smell

The vibrational theory of olfaction explains several aspects of odorant detection that theories based purely on receptor binding do not. It provides for additional selectivity through receptors that are tuned to specific vibrational bands of the odorants they bind, and also through the subsequent conduction of electrons across the odorant, presumably by a tunneling mechanism. A lot of people seem like the theory, or at least its main theorist, Luca Turin. Over the years, efforts to prove, or disprove the vibrational theory have progressed through a long series of olfactory touchstones: molecular enigmas like carvone, acetophenone, or benzaldehyde, whose experimentally perceived scent is a seemingly fickle amalgam of various molecular vibrations, mirror images, and isotopes.
The latest assault designed to topple the theory comes from Eric Block of State University of New York, and several of his colleagues. Their new results, published today in PNAS, focus on a peculiar class of odorants collectively know to us as musks. The power of some of these musks, like those related to civetone, are well known to hunters and photographers who have been said to use Calvin Klein's Obsession perfume to lure big cats. Taking a cue from Turin's own suggestions and experiments that humans could distinguish large musk molecules whose vibration frequencies were altered in a sufficient number of places by substituting deuteriums for hydrogens, Block set out to see whether the actual receptors might pass similar judgment.
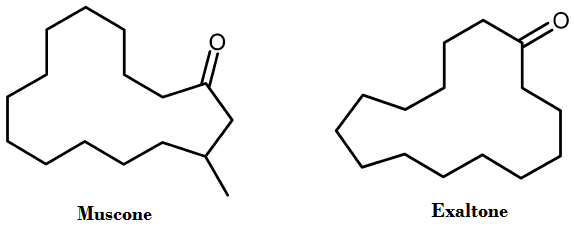
To do this his group synthesized a panel of various 'isotopomers' of different musks, in particular, Exaltone® (cyclopentadecanone) and muscone. Isotopomers are simply isotopic isomers—ie. isomers with isotopic atoms, having the same number of each isotope of each element but differing in their positions. The researchers identified a putative human musk olfactory receptor, OR5AN1, and found that all of their isotopomers gave similar receptor responses. They also looked at the responses of a homologous mouse musk receptor, MOR244-3, to their isotopomer panel as well as to deuterated and nondeuterated acetophenone and benzaldehyde, and to selected 13C isotopomers.
The way researchers typically evaluate olfactory receptor responses is to express them in either oocytes or kidney cells, and then measure any electrical or chemical response to application of the odorant. Here the researchers use a luciferase reporter assay to measure response elements like c-Fos, which are activated by cAMP. As G-protein coupled receptors (GPCRs), olfactory receptors generate cAMP upon activation. Compared to direct electrical effects, the cAMP route (and especially any subsequent gene induction), would in turn be expected to be a much slower response.
After making various NMR and IR spectrographic measurements of some sample odorants, the researchers suggest that Turin's results may be due either to impurities, or to extra-receptor events. They also claim that their finding with musks, and a few other odorants casts doubt on the entire vibrational foundation. Turin, in the meanwhile, has moved on to solving other unanswered biophysical conundrums, like for example, the more general question of electron transport in proteins. In particular, as it pertains to other esoteric phenomena like electron spin effects in anesthesia. Naturally enough, Turin has a few objections to these claims, both with respect to the physical mechanisms and the biological mechanisms under interrogation here.
Regarding the physics aspects Turin notes,
"I have asked Klaus Schulten, Ilya Solov'yov, Andrew Horsfield and Eric Bittner whose work is discussed in the article to comment. Their unanimous reaction was that they were well aware of the problems raised by Block et al. and had in fact discussed them at length in their publications. All feel that a definite conclusion must await a proper structure for an olfactory receptor."
As to the biology, Turin pointed out several contraindications which I will just list below;
1- An obvious question that they do not touch upon is: do the deuterated musks smell different? If not, why did they not mention it since it would have been strong evidence against our previous paper. If yes, surely they should mention it. Either way, we would be grateful for some samples for evaluation, or of course we could make our own.
2- A negative in vitro result is hard to compare with a positive in vivo one:
- We have no idea whether these olfactory receptors expressed in kidney cells work identically to the native ones or whether they have been altered/damaged by heterologous expression. There are many ways of getting a negative result, not all of which are interesting.
- Receptor activation in the vibrational theory requires two steps: A- binding, and B- a match of the band perceived by the receptor to vibrations of the odorant. Block and colleagues do not know whether the receptors they are looking at sense the correct band.
3 -They make much of differences between deuterium and hydrogen: "Thus, the acidity of D2O and H2O are different, hydrogen bonding of O–H and O–D bonds are different, boiling points and freezing points are different, etc. In particular, the gas chromatographic retention times of isotopomeric pairs in the present study are significantly different in all cases examined."
- True, but none of these things are related in any way to odorant-receptor interactions, since the deuteriums they introduce are non-exchangeable, i.e. not acid, do not form hydrogen bonds, and the boiling and freezing points are irrelevant as of course is retention time in a chromatography column.
- Furthermore, all their results contradict the above, since they show in all cases that the deuterated odorant have identical affinity to the receptor.
More information: Implausibility of the vibrational theory of olfaction , PNAS, www.pnas.org/lookup/doi/10.1073/pnas.1503054112
Journal information: Proceedings of the National Academy of Sciences
© 2015 Phys.org



















