March 5, 2015 feature
Buckybomb shows potential power of nanoscale explosives
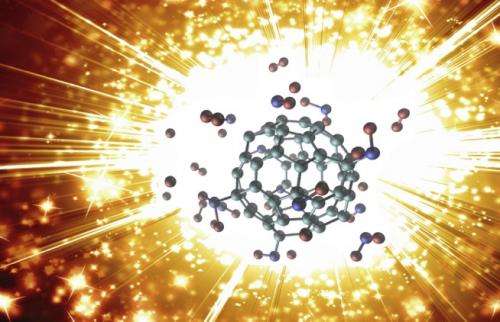
(Phys.org)—Scientists have simulated the explosion of a modified buckminsterfullerene molecule (C60), better known as a buckyball, and shown that the reaction produces a tremendous increase in temperature and pressure within a fraction of a second. The nanoscale explosive, which the scientists nickname a "buckybomb," belongs to the emerging field of high-energy nanomaterials that could have a variety of military and industrial applications.
The researchers, Vitaly V. Chaban, Eudes Eterno Fileti, and Oleg V. Prezhdo at the University of Southern California in Los Angeles, have published a paper on the simulated buckybomb explosion in a recent issue of The Journal of Physical Chemistry Letters. Chaban is also with the Federal University of São Paulo, Brazil.
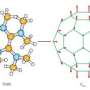
The buckybomb combines the unique properties of two classes of materials: carbon structures and energetic nanomaterials. Carbon materials such as C60 can be chemically modified fairly easily to change their properties. Meanwhile, NO2 groups are known to contribute to detonation and combustion processes because they are a major source of oxygen. So, the scientists wondered what would happen if NO2 groups were attached to C60 molecules: would the whole thing explode? And how?
The simulations answered these questions by revealing the explosion in step-by-step detail. Starting with an intact buckybomb (technically called dodecanitrofullerene, or C60(NO2)12), the researchers raised the simulated temperature to 1000 K (700 °C). Within a picosecond (10-12 second), the NO2 groups begin to isomerize, rearranging their atoms and forming new groups with some of the carbon atoms from the C60. As a few more picoseconds pass, the C60 structure loses some of its electrons, which interferes with the bonds that hold it together, and, in a flash, the large molecule disintegrates into many tiny pieces of diatomic carbon (C2). What's left is a mixture of gases including CO2, NO2, and N2, as well as C2.
Although this reaction requires an initial heat input to get going, once it's going it releases an enormous amount of heat for its size. Within the first picosecond, the temperature increases from 1000 to 2500 K. But at this point the molecule is unstable, so additional reactions over the next 50 picoseconds raise the temperature to 4000 K. At this temperature, the pressure can reach as high as 1200 MPa (more than 10,000 times normal atmospheric pressure), depending on the density of the material.
Chemically speaking, the scientists explain that the heat energy comes from the high density of covalent energy stored by the carbon-carbon bonds in the C60. Because the NO2 groups initiate the reaction, adding more NO2 groups increases the amount of energy released during the explosion. Choosing an appropriate number of these groups, as well as changing the compound concentration, provide ways to control the explosion strength.
The researchers predict that this quick release of chemical energy will provide exciting opportunities for the design of new high-energy nanomaterials.
More information: Vitaly V. Chaban, et al. "Buckybomb: Reactive Molecular Dynamics Simulation." The Journal of Physical Chemistry Letters. DOI: 10.1021/acs.jpclett.5b00120
Journal information: Journal of Physical Chemistry Letters
© 2015 Phys.org