Researchers report better solar cells through chemistry
In the quest for the perfect solar cell – efficient, thin, reliable and cheap – new Cornell research offers quantifiable insight into the complex chemistry of getting it just right.
A research team that includes Uli Wiesner, the Spencer T. Olin Professor of Materials Science and Engineering, and Lara Estroff, associate professor of materials science and engineering, has demonstrated how to optimize the fabrication conditions of a thin film solar cell widely believed to show promise for photovoltaic applications. Their work was published Jan. 30 in Nature Communications.
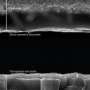
Thin film solar cells are made by depositing a thin layer of photovoltaic material onto a substrate, such as silicon. The type of films the Cornell researchers made are organic-inorganic metal halide perovskites, which have been widely studied for solar applications over the last several years, according to Wiesner.
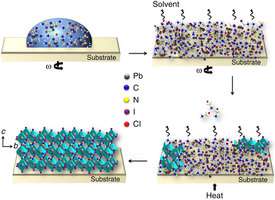
The researchers arrived at near-perfect solar cells using a new liquid source, called a non-halide lead acetate. While perovskite solar cells have improved rapidly in a few short years, some basic properties of the light-absorbing material still weren't well understood. Making the solar material starts with a solution of organic and inorganic molecules, and tuning the solution for smooth, defect-free perovskite films hadn't been perfected.
The researchers started with several kinds of liquid sources made of lead salts and studied the role that certain negatively charged ions - anions - played in the film formation. With the help of the Cornell High Energy Synchrotron Source, they characterized how the perovskites' crystal structures varied depending on which lead salt solution was used. From there they could figure out which was optimal.
The method they settled on for growing the solar cells is also a simple solution-coating, which makes it particularly appealing for commercial applications.
"By choosing the right precursor chemistry and via a simple one-step solution casting process, we obtained perovskite films with a smoothness surpassing that of vapor-deposited films, resulting in record power conversion efficiencies," Wiesner said.
The paper, "Ultrasmooth Organic-Inorganic Perovskite Thin-Film Formation and Crystallization for Efficient Planar Heterojunction Solar Cells," includes work at four universities on three continents: Cornell, with Oxford University and Cambridge University in the United Kingdom, and Nanjing University in China. It is the result of a longstanding collaboration between the Wiesner and Estroff labs at Cornell, and their interaction with Henry Snaith at Oxford University, a world leader in perovskite solar cell device fabrication.
More information: "Ultrasmooth organic–inorganic perovskite thin-film formation and crystallization for efficient planar heterojunction solar cells." Nature Communications 6, Article number: 6142 DOI: 10.1038/ncomms7142
Journal information: Nature Communications
Provided by Cornell University