January 22, 2015 feature
Reversible solid-to-liquid phase transition offers new way to synthesize crystals
(Phys.org) —The simple acts of heating and cooling affect different substances in different ways: some substances may change phase from solid to liquid to gas, while others may irreversibly break down when heat is applied. In a new study, scientists have investigated how changes in temperature affect a class of inorganic-organic hybrid crystals called coordination polymers (CPs), and show that, for some of them, the reversible solid-to-liquid phase transition can serve as an alternative way to synthesize and process these valuable materials.
The researchers, led by Satoshi Horike at Kyoto University and PRESTO (the Japan Science and Technology Agency) and Susumu Kitagawa at Kyoto University, have published their paper on reversible phase transitions in CP crystals in a recent issue of the Journal of the American Chemical Society.
The general molecular structure of CPs consists of a central metal ion surrounded by organic molecules called ligands. These units are then bonded together by the ligands to form large networks. This arrangement leads to a variety of interesting magnetic, optical, and electrical properties. As a result, CPs have many diverse applications, such as in dyes, lighting, gas storage, and catalysis.
So far, however, not much is known about the reversible solid-to-liquid phase transition of these materials, which is partly due to the fact that most CPs do not melt but irreversibly decompose when heated. When a substance melts, the bonds between molecules break, whereas when a substance decomposes, the bonds within each individual molecule break. These intramolecular bonds are not usually reformed by lowering the temperature, and so the decomposed substance does not re-solidify.
"Inorganic-organic hybrid CPs (including metal organic frameworks, MOFs) have been studied intensively from the early 1990s, and so far over 20,000 compounds have been reported," Horike told Phys.org. "However, there are few reports on 'melting" behavior' – a fundamental thermal process. In this work, we demonstrated the synthesis of melting CPs and how they melt by various characterizations. The significant points of this work are, first, the discovery of a stable liquid state in some CPs. Most researchers in CPs and MOFs have worked only on solid states. And second, we suggest a new approach of material design of CPs by use of their liquid state."
As he explained, CPs that can melt are unusual because of their stability.
"There is always competition between 'structure decomposition' and 'formation of a stable liquid state' for CPs at high temperature," Horike said. "Usually, CPs start to decompose as temperature increases because, once coordination bonds break, then immediately organic ligands start to decompose or vaporize. The melting CPs have stable liquid states below the decomposition temperature."
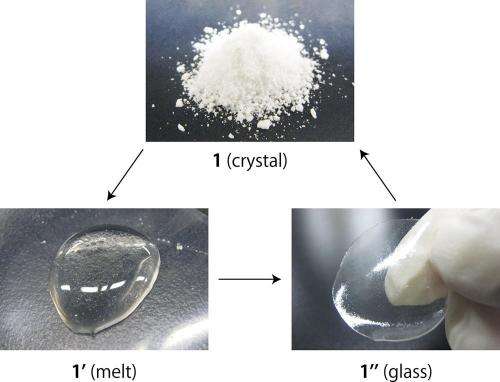
In their study, the researchers performed experiments in which they melted a few of the CPs that do actually melt instead of decompose. The researchers found that, even though the polymers' bonds are not fully preserved in the liquid state, they are fully reformed in the solid state, causing the liquid to recrystallize as a glass. The scientists explain that reforming of the bonds is possible partly due to the materials' high viscosity, along with a fine balance of their other properties.
Using this melting behavior, the scientists showed how selected CP crystals can be fabricated with a melt-growth technique. They demonstrated that a melted CP could slowly recrystallize into a crack-free crystal. Such a crystal can form even in a narrow space, which is not possible with conventional crystallization methods. This new fabrication technique has the potential for use in the many diverse applications of CPs.
"We can expect many things," Horike said. "For example, CPs have gas separation or catalysis properties, and these functions require morphology control or shaping of solids such as fabrication of films, fibers, and composites. We can fabricate them through their melting behavior and it is very easy, homogenous, and on a large scale. Conventionally, researchers need to control the kinetics of crystal growth in solution, or use a top-down approach (such as ball milling) to downsize the particles of CPs.
"We also expect that the liquid CPs could be regarded as a new class of functional liquid (though it is related to ionic liquids), and the liquid CPs could be available for catalysis, ion conduction/dielectricity, magnetic fluids, and other uses."
More information: Daiki Umeyama, et al. "Reversible Solid-to-Liquid Phase Transition of Coordination Polymer Crystals." Journal of the American Chemical Society. DOI: 10.1021/ja511019u
Journal information: Journal of the American Chemical Society
© 2015 Phys.org