Enhancing microbial pathways for biofuel production

Researchers from the DOE JGI and the Joint Bioenergy Institute identified genes in an E. coli microbial metabolism pathway that could improve the production of terpenes.
Terpenes are high-energy compounds produced in microbes and plants that could be used for producing biofuels. Enhancing terpene yields could lead to commercial-scale production of these biofuels.
Terpenes are hydrocarbons in plants such as conifers that act as a self-defense mechanism against pests, among other functions. Bioenergy researchers see terpenes are high-energy metabolites that could be used for producing biofuels from plant feedstocks. For example, terpene production in eucalyptus is of interest to the bioenergy researchers who were part of an international consortium of researchers, including scientists from U.S. Department of Energy Joint Genome Institute (DOE JGI), a DOE Office of Science user facility, which described the eucalyptus genome in Nature earlier this year. One side project resulting from work done there is being led by study co-author Jerry Tuskan of the DOE JGI as well as Oak Ridge National Laboratory (ORNL) and the BioEnergy Science Center (BESC), a DOE Bioenergy Research Center (BRC). Tuskan's team is working on oil gland formation in plants and expressed interest in determining the biochemical pathway of terpene production in eucalyptus leaves to develop a sustainable alternative to jet fuel.
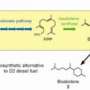
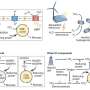
In the January 2015 issue of Applied and Environmental Microbiology, DOE JGI researchers collaborated with another BRC, the Joint BioEnergy Institute (JBEI), to find ways of enhancing terpene yield in bacteria. In previous studies, JBEI researchers had reported that bisabolane, a biofuel resulting from the precursor terpene bisabolene, could serve as an alternative to diesel fuel. They wanted to find a way to improve terpene production in E. coli using the metabolic DXP pathway, which they consider more efficient in terms of final yield compared to the mevalonate pathway. To develop a novel route that would take C5 sugars (such as the xylose formed when hemicellulose is broken down) to terpenes, they used a directed-evolution strategy and deleted specific genes involved a key point in the pathway. The results led to their discovery of two novel routes: one that arose through spontaneous mutations; and, one found through overexpression of a selected candidate gene, for producing the terpene and candidate biofuel bisabolene.
The team also noted that applying the engineering process to the DXP pathways in plants and algae "could provide a more direct link from carbon fixation (Ru5P in the Calvin cycle) to the terpene pathway."
More information: Kirby J et al. "Enhancing Terpene Yield from Sugars via Novel Routes to 1-Deoxy-d-Xylulose 5-Phosphate." Appl Environ Microbiol. 2015 Jan 1;81(1):130-8. DOI: 10.1128/AEM.02920-14
Journal information: Applied and Environmental Microbiology
Provided by US Department of Energy