Geochemical reactions may decrease effectiveness of carbon storage schemes
New research shows that the natural reactions taking place in some of the underground reservoirs used to store carbon dioxide may prevent carbon emissions from being transported to greater depths, where it may be less likely to leak into the atmosphere.
Geochemical reactions taking place in aquifers – underground layers of water-bearing porous rock –may lead to carbon dioxide being 'pooled' for hundreds or even thousands of years, and may force a rethink of how these underground reservoirs are used in carbon capture and storage (CCS) schemes.
The new research, from the University of Cambridge, has shown that aquifers rich in silicate minerals may delay, or even prevent, CO2 from being carried to greater depths, where it may be less likely to leak out of the aquifer and into the atmosphere. The results are published in the journal Nature Communications.
Saline aquifers have been considered the safest and most efficient option for CCS schemes, where anthropogenic carbon emissions are trapped and stored underground so that they do not enter the atmosphere.
Both dissolution in the formation water and transport to depth decrease the risk of CO2 escaping: dissolution reduces the risk of potential upward leakage through fractures in the cap rock of the reservoir, while transport to greater depth increases the rate of dissolution and of potential incorporation of the CO2 into the rock minerals.
"Our research has found that CO2 may not behave as expected when stored in aquifers, challenging some of our previous assumptions about CCS schemes," said Dr Silvana Cardoso of the Department of Chemical Engineering and Biotechnology, who led the research.
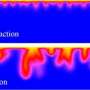
It has been thought that once the CO2 is dissolved in the aquifer water, making it denser, convection streams develop and carry the mixture to deeper parts of the aquifer.
Dr Cardoso and her co-author, Jeanne Andres, a former PhD student in the same department, found that chemical reactions between the rock formations and the dissolved CO2 may delay, or even prevent, the CO2 from reaching greater depths by decreasing the strength of the convection streams.
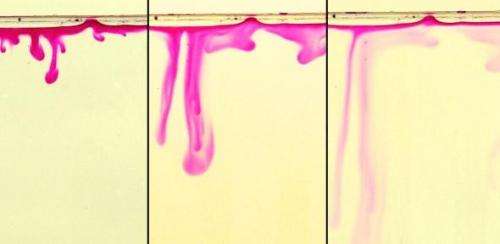
The researchers used a combination of simple laboratory experiments and mathematical analysis to establish the basic interaction between fluid flow and chemical kinetics in a deep porous medium. Their study assessed the impact that the natural chemical reactions between the dissolved CO2 and the rock formation have on the convection streams which carry the CO2 to greater depths.
The researchers found that the behaviour of carbon dioxide depends strongly on the chemical composition of the rock formation: while the streaming of dissolved carbon dioxide persists in carbonate rocks, the chemical interactions in silicate-rich rocks may curb this transport drastically and even inhibit it altogether. For example, for a rock matrix rich in calcium feldspar, the convection streams may be completely shut off just two months after the onset of motion. After this, the carbon dioxide will be transported to depth by much slower diffusional processes.
These results challenge current views of carbon sequestration and dissolution rates in the subsurface, suggesting that pooled carbon dioxide may remain in the shallower regions of the formation for hundreds to thousands of years, while deeper regions of the reservoir can remain virtually carbon free.
The findings could have important practical implications for storage of carbon dioxide in saline aquifers, enabling informed screening of the most effective sites.
"Screening of new sites will need to include not only the size and location of the reservoir, but also the mineralogy of the rock," said Cardoso. "The present study simply shows that mineralogy has a strong impact on where the CO2 ends up. Which specific mineralogy is best remains to be studied."
More information: "Geochemistry of silicate-rich rocks can curtail spreading of carbon dioxide in subsurface aquifers." Nature Communications 5, Article number: 5743 DOI: 10.1038/ncomms6743
Journal information: Nature Communications
Provided by University of Cambridge