New form of ice could help explore exciting avenues for energy production and storage
Clathrates are now known to store enormous quantities of methane and other gases in the permafrost as well as in vast sediment layers hundreds of metres deep at the bottom of the ocean floor. Their potential decomposition could therefore have significant consequences for our planet, making an improved understanding of their properties a key priority.
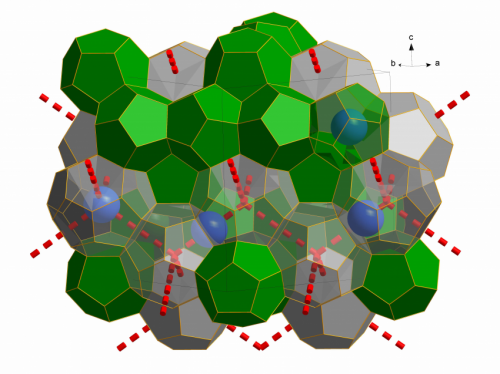
In a paper published in Nature this week, scientists from the University of Göttingen and the Institut Laue Langevin (ILL) report on the first empty clathrate of this type, consisting of a framework of water molecules with all guest molecules removed. Long thought to be purely hypothetical, this empty clathrate plays an important role in our understanding of the physical chemistry of gas hydrates. Such research could help ease the flow of gas and oil through pipelines in low temperature environments, and open up untapped reservoirs of natural gas on the ocean floor.
In order to create the sample of Ice XVI, the researchers constructed a clathrate filled with molecules of neon gas, which they then removed by careful pumping at low temperatures. Using small atoms such as those of neon gas allowed the clathrate to be emptied without compromising its fragile structure.
In order to achieve this, the neon clathrate was pumped in a vacuum at temperatures in the region of 140° K, while neutron diffraction data was taken using the ILL's state-of-the-art D20 diffractometer. The images obtained allowed them to confirm when the clathrate had been fully emptied, and provided a complete picture of the resulting structure.
As a stable solid composed entirely of molecules of H2O, the empty clathrate also represents a new phase of ice. Ice XVI is the 17th discovered form of ice, and is the least dense of all known crystalline forms of water. It is also predicted to be a stable low-temperature configuration of water at negative pressures (the equivalent of tension - the opposite of compressive positive pressures), and is so far the only experimentally-obtained form of ice to have a clathrate configuration.

As the empty clathrate is used as a reference frame for numerous molecular simulations, scientists have up until now relied on approximate theoretical models to underpin their work. The empty clathrate framework obtained at the ILL will now allow for the accurate determination of its fundamental structural and thermodynamic properties. The ability to create and observe such empty clathrates has the potential to considerably improve our understanding of these compounds when they are filled with gas.
According to the 2007 World Energy Outlook, the total amount of methane stored within clathrates on the ocean floor far exceeds the economically exploitable reserves of "conventional" carbon in the form of coal, petrol or natural gas left on Earth. These reservoirs are difficult to exploit at present but form a domain of intense ongoing research.
Thomas Hansen, one of the study authors and instrument scientist on D20 at the ILL, says: "It is important to note that clathrates could also be formed with carbon dioxide gas which would create stable compounds on the ocean floor. This means there is a possibility we could extract methane and convert it to useful energy, and replace it with the CO2. In other words, we could pump CO2 down to the ocean floor as a replacement for the methane in the gas hydrates. The challenges involved would naturally be large and the feasibility has been called into question, but it remains an extremely intriguing possibility worth exploring.
Hansen adds: "My co-authors Andrzej Falenty and Werner F Kuhs from the University of Göttingen are both members of the SUGAR project funded by the German government, whose aim is to explore the scientific, technical and economic possibilities of such an undertaking. Similar activities are currently underway in Japan, China, India and elsewhere."
Helmut Schober, ILL Science Director, said: "Empty clathrate hydrates have for many years been the subject of intense scientific speculation, as their very existence was relatively uncertain. The present discovery definitively resolves this speculation, and provides us with a new jewel in the fascinating treasure chest of ice phases. Predicting the properties of ice XVI will be a benchmark for any model that pretends to describe the physics of water. This in itself is a tremendous step forward. It is via this understanding that we hopefully will also advance on the surrounding questions related to energy."
An area where clathrate research is of more immediate benefit is in the maintenance of pipelines where gas is transported at high pressures and low temperatures. These conditions can lead to the production of gas hydrates within the pipes, which in turn form substantial blockages, the prevention of which costs industry approximately $500 million a year worldwide. Given the high international economic impact of such pipelines, this represents a significant cost factor that further research on clathrate properties can help reduce.
More information: Formation and properties of ice XVI obtained by emptying a type sII clathrate hydrate, Nature, DOI: 10.1038/nature14014
Journal information: Nature
Provided by Institut Laue-Langevin