Molecules in a fluid not randomly arranged
FOM PhD researcher Matthijs Panman and his colleagues from the University of Amsterdam have demonstrated that molecules in liquid alcohol are not randomly oriented with respect to each other. The angle between the oxygen-hydrogen bonds of two neighbouring alcohol molecules is usually about 120 degrees. This discovery refutes the commonly held idea that molecules in a liquid are randomly arranged. The researchers published their work on 12 November 2014 in Physical Review Letters.
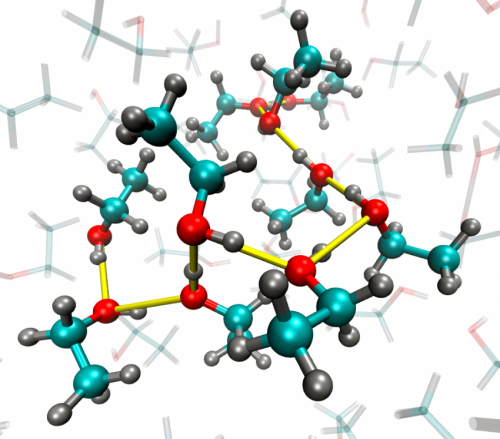
In school we learn that molecules in a liquid are randomly arranged. But is that completely correct? Based on computer simulations it has been thought for many years that the molecules in many liquids are locally ordered, especially if the molecules have a strong interaction with each other as is the case for water or alcohol. Panman has shown that this idea is correct: in alcohol there is local orientational order. This means that nearby molecules are ordered at a fairly well-defined angle with respect to each other, although there is no long-distance ordering like in a crystal.
Light and vibrations
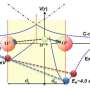
The FOM researchers, working at the Van 't Hoff Institute for Molecular Sciences in Amsterdam and in collaboration with the Amsterdam Center for Multiscale Modeling, devised an experiment to observe the local ordering. Each alcohol molecule contains a bond between its oxygen atom (O) and hydrogen atom (H). The researchers illuminated alcohol with infrared light pulses with a duration of about one trillionth (10-12) of a second. If the infrared light has the right frequency, some of the O-H bonds in the alcohol molecules will start to vibrate as a result of resonance. The bond becomes longer and shorter synchronously with the light wave.
The light used is polarised, which means that the electric field of the light has a fixed direction. The light only causes the O-H bonds that are parallel to its electrical field to vibrate. Subsequently these vibrating bonds pass on the movement to O-H bonds in neighbouring molecules. This energy transfer process is possible because all the O-H bonds have the same resonance frequency. If two neighbouring O-H groups are not parallel to each other but at an angle, then after the transfer of the vibration, O-H bonds molecules that are not parallel to the electrical field of the infrared pulse will also vibrate. The strength of the effect (the loss in directionality) depends on the angle between the O-H bonds of the neighbouring molecules.
120 degrees
By calibrating the transfer of vibrations to predictions of this rate from their computer simulations of the liquids, the researchers could estimate the average angle between neighbouring molecules. They discovered that neighbouring O-H bonds in alcohol were at an angle of about 120 degrees with respect to each other. In other liquids that angle can be different: for example in the liquid N-methylacetamide the bonds between the nitrogen atom and the hydrogen atom of neighbouring molecules are roughly parallel with each other.
The local order in both liquids is caused by interactions (hydrogen bonds) between the neighbouring molecules. The local order only exists for several trillionths (10-12) of a second, because the network of hydrogen bonds is highly dynamic: hydrogen bonds between molecules constantly break and form.
The research provides a better understanding of liquids and the behaviour of their molecules. A good knowledge of liquids is vital to understand how chemical reactions take place, so that the efficiency of these reactions can be increased.
More information: Local Orientational Order in Liquids Revealed by Resonant Vibrational Energy Transfer, Phys. Rev. Lett. 113, 207801 – Published 12 November 2014. journals.aps.org/prl/abstract/ … ysRevLett.113.207801
Journal information: Physical Review Letters
Provided by Fundamental Research on Matter (FOM)