Better diagnostic imaging for traumatic brain injuries

Image-calibration technology designed and developed by scientists at the National Institute of Standards and Technology (NIST) in collaboration with the National Cancer Institute and the Radiological Society of North America has been adopted for use in multi-site clinical trials in the United States and Europe to study the effects of traumatic brain injury (TBI).
By mid-November of 2014, about two dozen of the units –which were brought from conceptualization to commercialization in only a year –will have been distributed to trial participants in an effort to bring uniform quality control to a critically important technique in brain imaging.
Recent publicity about concussions and other head injuries in the National Football League and among soldiers exposed to explosions has raised awareness of TBI. But the condition is by no means restricted to professional sports and military service. TBI is a major cause of death and disability in the United States, contributing to about 30% of all injury deaths, according to the Centers for Disease Control and Prevention.
The principal diagnostic tool for detecting and monitoring microscopic changes caused by TBI is magnetic resonance imaging (MRI). But MRI scanners are made by several manufacturers, operated in a variety of clinical settings, and generate images using different values for key parameters. To ensure that brain and other scans are not only accurate but also comparable to images of the same patient taken at different times, or to images made on other scanners, physicians need a quantitatively consistent standard against which to calibrate their instruments.
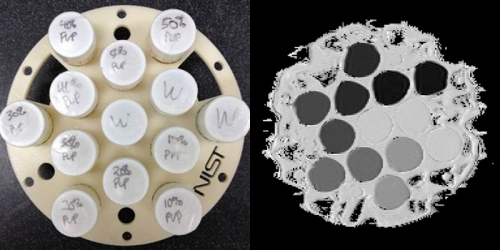
Providing such standards is a long-term goal of researchers at the Physical Measurement Laboratory's Magnetics Group. Over the past 12 months, in response to requests from clinicians, a team from that group conceptualized and made prototypes of a head "phantom" (calibration standard) the size of a volleyball. It is filled with containers of polymer solutions at various carefully prepared concentrations. These serve as standard surrogates for water having different rates of diffusion in the brain.
Water diffusion is a major indicator of injury in TBI, and "it can be used to reveal conditions that otherwise would be invisible," said Michael Boss of the Biomagnetic Imaging Standards and Microsystems program.
"Neural fibers in your brain are basically like a bundle of wires, and in a concussive shock those wires can break," Boss said. "Water tends to diffuse along the direction of the neural bundle. Any disruption in neural connections is revealed in a corresponding change in diffusion pattern, giving a map of the affected area. But doctors need to be sure that they're measuring diffusion correctly, and that's where our diffusion phantom comes into play."
The phantoms provide "ground truth" values for diffusion in the form of about a dozen vials, each containing a different concentration of a polymer called PVP dissolved in water, mounted within a spherical plastic shell. Because diffusion is extremely sensitive to temperature, MRI scanners operating at even slightly different temperatures will give different readings for the same concentration of PVP. To minimize that problem, the NIST-designed phantoms are cooled by a unique ice-water bath system so that different users will make measurements of the same property of interest at the same temperature: 0 ͦ C.
In the fall of 2013, initial prototypes of the phantoms were circulated for testing and evaluation to collaborators in the IMI-QuIC-ConCePT initiative in the European Union. Early this year, a prototype was shipped to a U.S. traumatic brain injury clinical trial called TRACK-TBI, supported by the National Institutes of Health.
By May, Boss and colleagues were well on the way to a final configuration. Adjustments in the phantom's plastic components and vial sizes eliminated artifacts in images. Switching to transparent polycarbonate made it much easier to remove air bubbles. Alterations in the shell design made it possible to add ice from both sides for more even cooling, while also maintaining a shape that was easily rotated and a size that would fit in the head coils of different MRI scanner manufacturers.
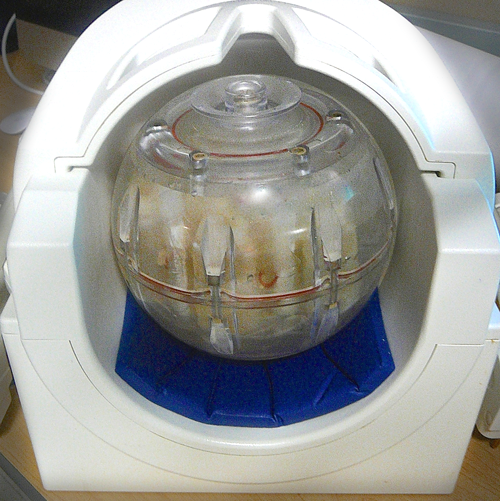
Once a final design was decided upon, the phantoms were commercialized by High Precision Devices of Boulder, Colorado. Those units are now being shipped to clinical sites in the United States and Europe.
The technology has utility beyond TBI, Boss said, and is adaptable to various kinds of imaging in other kinds of neurotrauma, cancer, stroke, and in the study of neurodegenerative diseases. The team is already at work on designs for those uses, as well as further improvements on the current diffusion phantom.
"The entire effort, from conception to commercialization, came together in an amazingly short period of time. The diffusion phantom built on our previous expertise in developing phantoms for entire MRI systems. We are now developing other MRI phantoms for specific modalities, such as one to calibrate coils used in clinical studies of breast cancer," said project leader Stephen Russek.
The Magnetics Group has a long history of cutting-edge research in high frequency magnetism. Group leader Ron Goldfarb noted that "phantom development is part of our larger program in magnetic resonance, which includes the development of new types of nano-agents for MRI contrast and cell tracking, and advances in low-field magnetic-resonance imaging methods and sensors. NIST's Physical Measurement Laboratory—in both Boulder, Colorado, and Gaithersburg, Maryland—has several programs devoted to methods for precise, calibrated measurements in medical physics."
Provided by National Institute of Standards and Technology



















