DARPA-funded DEKA arm system earns FDA approval
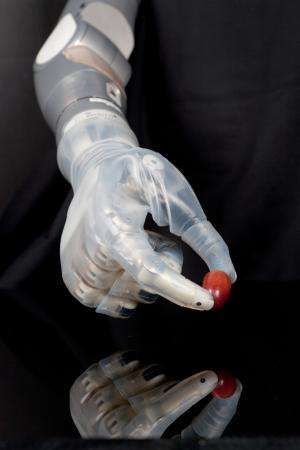
DARPA launched the Revolutionizing Prosthetics program with a radical goal: gain U.S. Food and Drug Administration (FDA) approval for an advanced electromechanical prosthetic upper limb with near-natural control that enhances independence and improves quality of life for amputees. Today, less than eight years after the effort was launched, that dream is a reality; the FDA approved the DEKA Arm System.
"DARPA is a place where we can bring dreams to life," said Dr. Geoffrey Ling, M.D., Director of DARPA's Biological Technologies Office . Dr. Ling is a retired Medical Corps neuro-critical care officer who served in combat hospitals in both Iraq and Afghanistan. He launched the Revolutionizing Prosthetics program in 2006 with a desire to provide better care "to repay some of the debt we owe to our Service members."
The newly approved system expands prosthetic choices for amputees, who have generally used body-powered prosthetics – in particular the split-hook device invented in 1912. DARPA took up the challenge to advance the field of modular upper-limb prosthetics and committed to making the significant research and development investment required.
The system allows for simultaneous control of multiple joints using a variety of input devices including wireless signals generated by innovative sensors on the user's feet. The battery-powered arm system is of similar size and weight to a natural limb and has six user-selectable grips. The DEKA Arm System is indicated for individuals 18 and older.
Many technological breakthroughs at the intersection of biology and engineering were necessary to enable the creation of the arm system: for example, miniaturization of parts for motors, computer controls and sensors, and manufacturing processes with lightweight, but strong materials.
A number of federal agencies contributed to the Revolutionizing Prosthetics program. The FDA reviewed information from a Department of Veterans Affairs-funded Optimization Study, which included data from 36 participants. The United States Army Medical Research and Materiel Command provided funding assistance to enable completion of tests and trials required to meet FDA requirements. The U.S. Army Research Office provided contract management support.
"Interagency collaboration has been critical to the Revolutionizing Prosthetics program to allow for independent assessments of the technology, incorporation of user feedback into the design, and identification of regulatory strategies and transition paths," said Dr. Justin Sanchez, the current program manager. "We could not have achieved our goal so quickly without the support of many partners in government."

The DEKA Arm System was developed by DEKA Integrated Solutions in Manchester, N.H. FDA approval allows DEKA to pursue manufacturing and commercial opportunities to bring the arm to market.
Provided by DARPA