New reactor enables immediate determination of optimum conditions for chemical reactions
Researchers of the University of Twente's MESA+ research institute have developed a new type of microreactor allowing for the targeted observation of chemical reactions by means of an electric field. The tiny reactor enables one to follow the reaction through the use of infrared spectroscopy, providing the researchers with an improved fundamental insight into the reaction and allowing them to determine the optimum process conditions. Arturo Susarrey-Arce will defend his doctoral thesis on 11 April.
Being able to properly research the ideal conditions for specific chemical reactions to take place is of great benefit to the chemical industry sector. Arturo Susarrey-Arce has developed a new type of microreactor at the University of Twente that enables such research.
This reactor gives researchers the opportunity to determine the influence of an external electric field on a chemical reaction. The use of such an electric field allows for, among other things, to tune reactivity during a chemical reaction and improving selectivity, resulting in, for example, a purer end product.
Microreactor

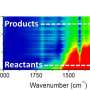
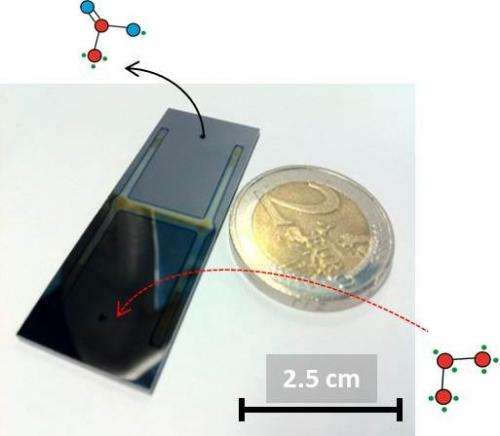
The tiny reactor - just a few centimeters in size - contains a thin, 900 nanometer to 2 micrometer-wide channel in which the reaction takes place. A variable electric field can be generated perpendicular to this channel to influence the reaction taking place within it. Next, using a specific type of infrared spectroscopy, researchers are able to, in situ, accurately observe the reaction while it is taking place. This means the microreactor can be utilized to determine the optimum process conditions of specific chemical reactions. Susarrey-Arce's doctoral research involved designing, manufacturing and analyzing the reactor, as well as proving its functional operability ('proof of principles'). In addition, he closely collaborated with the University of Twente's Physics of Fluids department to study the interaction between fluids and the walls of his reactor.
Provided by University of Twente