Understanding the energy and charge transfer of ions passing through membranes
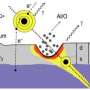
A bullet fired through a block of wood will slow down. In a similar way, ions are decelerated when they pass through a solid material: the thicker the material, the larger the energy loss will be. However, this picture breaks down in ultra-thin target materials, which only consist of a few layers of atoms.
Passing through a carbon membrane, highly charged ions can lose energy and change their charge state. If the case of extremely thin nano-membranes, however, two very distinct charge distributions are visible. They can be attributed to two different electron capture scenarios. Close collisions of the incident ion with target atoms can change the charge state dramatically and lead almost to neutralization, accompanied by considerable energy loss. If the ion, on the other hand, passes the target atoms at larger distances, it only captures a small number of electrons and hardly loses any energy. Therefore, the energy deposition by an ion in extremely thin materials cannot be expressed in terms of an average energy loss per ion path length.
These experiments show for the first time that the energy loss is strongly dependent on the charge state of the incident ion. Usually, this effect is not visible: in thicker films, the ions quickly reach an equilibrium charge state, independent of their original charge.
Understanding the energy and charge transfer of ions passing through membranes is a vital prerequisite for developing surface nanostructuring techniques by slow highly charged ions. The localized energy deposition in inelastic collisions with the target atoms makes it possible to engineer molecular sieves in a very simple and efficient way.

More information: Richard A. Wilhelm, Elisabeth Gruber, Robert Ritter, René Heller, Stefan Facsko, and Friedrich Aumayr. "Charge Exchange and Energy Loss of Slow Highly Charged Ions in 1 nm Thick Carbon Nanomembranes." Phys. Rev. Lett. 112, 153201 – Published 14 April 2014. dx.doi.org/10.1103/PhysRevLett.112.153201
Journal information: Physical Review Letters
Provided by Vienna University of Technology