March 17, 2014 report
Study shows short peptides can self-assemble into catalysts
(Phys.org) —A team of researchers with members from Syracuse University and the University of California, has found that naturally forming peptides can self-assemble into catalysts. In their paper published in the journal Nature Chemistry, the team describes how they designed seven peptides, allowed the resulting molecules to self-assemble into amyloids and then noted how many of them could catalyze the hydrolysis of esters.
Scientists have long wondered how enzymes could have possibly come to exist in the primordial soup that existed on Earth so long ago, leading to life as we know it. Enzymes, necessary for metabolic reactions, are made of large complicated (folded into three dimensions) proteins, with long chains of amino acids. That they could self-assemble into the structures we see today seems nearly impossible, even given millions of years (leading to the belief by some of the idea of intelligent design). Yet, life clearly exists, which means something, if not the hand of God, had to have happened to get them started. In this new effort, the researchers wondered if perhaps a simpler structure might have come to exist first, which then might have served as a step towards the development of enzymes. In this case, they wondered if such structures might have been amyloid plaques—the same type of molecule clusters found in the brains of Alzheimer's patients. Amyloids are much simpler than enzymes—so much so that most would believe they could come to exist without a guiding hand from an ethereal source.
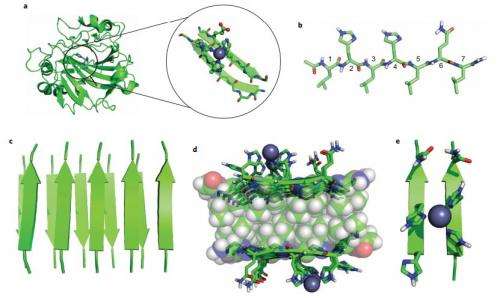
To learn more, the team created seven modest peptides, each of which were constructed from just seven amino acids—putting them together in a dish, with a dash of zinc iron to help move things along, allowed the peptides to self-form into different sheet-like fibril amyloids. During testing, four of the amyloids the team created were found to be able to catalyze the hydrolysis of esters.
The results of the team's experiments suggest that enzymes didn't necessarily have to spring forth fully formed from the primordial soup for life to begin, perhaps amyloids came first, serving as an intermediary, or a sort of blueprint—over millions of years the amyloids could have given way to the much more complicated enzymes, leading eventually to living organisms.
More information: Short peptides self-assemble to produce catalytic amyloids, Nature Chemistry (2014) DOI: 10.1038/nchem.1894
Abstract
Enzymes fold into unique three-dimensional structures, which underlie their remarkable catalytic properties. The requirement to adopt a stable, folded conformation is likely to contribute to their relatively large size (>10,000 Da). However, much shorter peptides can achieve well-defined conformations through the formation of amyloid fibrils. To test whether short amyloid-forming peptides might in fact be capable of enzyme-like catalysis, we designed a series of seven-residue peptides that act as Zn2+-dependent esterases. Zn2+ helps stabilize the fibril formation, while also acting as a cofactor to catalyse acyl ester hydrolysis. These results indicate that prion-like fibrils are able to not only catalyse their own formation, but they can also catalyse chemical reactions. Thus, they might have served as intermediates in the evolution of modern-day enzymes. These results also have implications for the design of self-assembling nanostructured catalysts including ones containing a variety of biological and non-biological metal ions.
Journal information: Nature Chemistry
© 2014 Phys.org