Nanoparticles with a core-shell structure can minimize the overheating of cells during bioimaging experiments
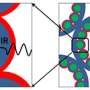
Upconversion nanoparticles—new types of luminescent nanomaterials that release high-energy photons after laser light stimulation—can penetrate deeper into tissue and are more photochemically stable than conventional bioimaging agents, such as quantum dots and organic dyes. Luminescent nanocrystals doped or impregnated with small amounts of rare-earth ytterbium (Yb) ions are particularly effective at photon upconversion. The specific lasers used to excite Yb dopants, however, can also heat water molecules in biological samples causing cell death or tissue damage.
Now, Xiaogang Liu from the A*STAR Institute of Materials Research and Engineering in Singapore and co-workers have synthesized a rare-earth-doped nanocrystal that can be excited at wavelengths within a safer 'biological window', thanks to a layered, core–shell design.
Luminescent nanocrystals require 'sensitizer' components to absorb photons and transfer energy to activator sites, which emit the desired light radiation. Liu and co-workers investigated a different rare-earth dopant, neodymium (Nd), which absorbs the short-wavelength laser light that excites water molecules, thus avoiding overheating effects. Unfortunately, Nd can be doped into nanocrystals only at very low concentrations before cross-interactions with activators begin to extinguish the luminescence. This makes Nd-doped nanoparticles weak emitters compared to Yb-based biomarkers.
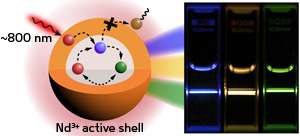
To resolve this problem, the researchers produced spherical nanoparticles containing layers with starkly different concentrations of Nd ions. They doped small amounts of Nd, Yb, and activator ions into nanocrystals of sodium yttrium fluoride (NaYF4), a material with a strong upconversion efficiency. They then synthesized a shell layer around the low-doped core containing a significantly higher Nd dopant concentration of 20 per cent. In this arrangement, the shell layer effectively harvests light and then transfers energy to the core, where low sensitizer concentrations minimize luminescence reduction (see image).
The experiments revealed that the core–shell design dramatically improved the nanocrystals' bioimaging capabilities—the new material had better light-harvesting capabilities than nanoparticles doped with pure Nd or Yb and achieved emission intensities seven times higher than pure NaYF4. Mechanistic studies showed that energy transfer between Nd and Yb ions in the nanoparticle core was key to overcoming the limitations of low dopant concentrations.
Next, the team tested their new materials by imaging an array of cervical cancer cells. While typical laser irradiation for Yb-doped biomarkers killed the cells within five minutes, the shorter wavelengths used for Nd-doped core–shell nanoparticles kept the cells viable over the same time.
"We plan to further improve the upconversion efficiency of our nanoparticles and use them for both bioimaging and drug delivery," says Liu.
More information: Xie, X., Gao, N., Deng, R., Sun, Q., Xu, Q.-H. & Liu, X. "Mechanistic investigation of photon upconversion in Nd3+-sensitized core–shell nanoparticles." Journal of the American Chemical Society 135, 12608–12611 (2013). dx.doi.org/10.1021/ja4075002
Journal information: Journal of the American Chemical Society