Plants in space: A novel method for fixing plant tissue samples maximizes time, resources, and data
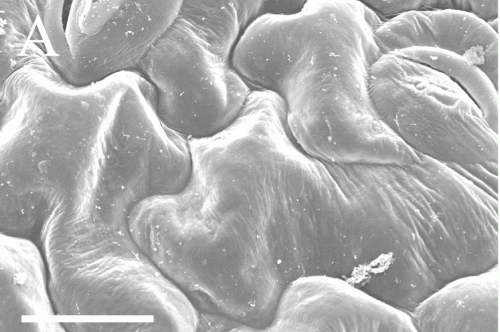
At work on the International Space Station, researchers studying plant and cell growth in space encountered a challenge. Imaging revealed interesting spaceflight-associated root morphologies. They needed to fix the tissues for further study back on Earth, but conventional fixation methods require separate fixatives depending on whether the sample is intended for molecular or morphological study. If the scientists wanted to study how spaceflight affected patterns of gene expression central to morphological patterns of cell growth, they needed a fixation method that would allow them to study both perspectives.
Most scientists at work in the laboratory rely on protocols that have been developed without the need for restrictions on the amount of space, time, or reagents they use. For scientists conducting experiments in spaceflight, time and resources are strictly regulated and limited, and researchers must know in advance which protocols will maximize the usefulness of the data they collect.
University of Florida professors Anna-Lisa Paul and Robert Ferl and colleagues are collaborating with the National Aeronautics and Space Administration (NASA) to understand plant growth and development in spaceflight. Along with lead author and graduate student Eric Schultz, they have developed a single fixation protocol for use in space that allows plant material to be used for multiple experimental applications. Their new protocol for sample preparation was tested on Arabidopsis tissues harvested on the International Space Station and is described in the August issue of Applications in Plant Sciences (available for free viewing at http://www.bioone.org/doi/pdf/10.3732/apps.1300034).
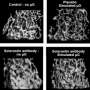
Because of limitations in astronaut crew time and orbital resources, previous spaceflight fixation protocols were designated as either molecular or morphological, due to the separate fixatives required for each application. Tissues for morphologic study were fixed in 3% glutaraldehyde (or a similar solution), and tissues for molecular study were fixed in the tissue storage reagent RNAlater. RNAlater has not commonly been used as a morphologic fixative, as it can produce unclear images with high background staining.
The new method developed by Schultz et al. puts RNAlater-fixed samples through a desalination process to return them to a fresh-like state, and then uses low-temperature scanning electron microscopy (cryo-SEM) to preserve tissues for imaging. Because few laboratories have access to the necessary equipment for cryo-SEM, the authors tested and developed a protocol that emulates cryo-SEM using standard SEM equipment and, importantly, that results in minimal tissue damage.
Although it was developed to address specific constraints for spaceflight experiments, Paul notes that their new method is broadly applicable. "There are a lot of situations where biologists want to collect samples in extreme situations. In our case—a space vehicle orbiting the Earth."
The new protocol maximizes the amount of data obtained from a single sample and allows for the concomitant examination of both molecular and morphological features. Using a single fixation protocol, direct comparisons between changes in morphology and altered gene expression can be made. Such an analysis not only makes full use of samples and replicates but also enables a robust analysis of the relationship between heredity and development. "Putting two tools together, it is powerful to look at the morphology in conjunction with the genes that are being expressed," says Paul.
The new protocol boasts low costs and high accessibility, and has wide application to any situation where recovery of biological resources is limited. Notably, this includes researchers collecting and preserving samples in the field, where space for materials is at a premium. "In places where sampling is limited, difficult, or expensive, the use of preservatives allows for more routes to analysis," notes Ferl.
Journal information: Applications in Plant Sciences
Provided by American Journal of Botany