September 6, 2013 report
New method of incorporating fluoride into drugs
(Phys.org) —Synthetic biology is a science that aims to harness natural biological processes for other uses, such as in the manufacture of fuels and drugs. Now, synthetic biology researchers in the U.S. have developed a new method of incorporating the highly reactive fluoride into a wider range of molecules than was previously possible.
Fluorine rarely occurs naturally in biological molecules, and many compounds containing fluorine are toxic. Fluorine is the most electronegative and reactive of all the elements, and it forms an extremely tight bond with carbon that is difficult for enzymes to break down. This can make it a useful additive for pharmaceutical chemicals, but fluorine's reactivity also makes it a difficult element to work with.
Fluorine is a common element added to pharmaceuticals because it can increase the drug's selectivity, enable it to dissolve in fats, and decrease the speed at which the drug is metabolized, thus allowing it more time to work. Twenty to thirty percent of drugs contain the element, including some of the most widely prescribed drugs such as well-known antibiotics, anti-inflammatories, anti-depressants, cholesterol-lowering medications, and some inhaled anesthetics.
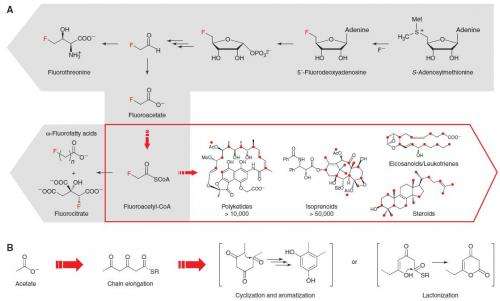
Until recently, the only known pathway for introducing fluorine into organic molecules was the fluoroacetate route. Now, Michelle C.Y. Chang and Mark C. Walker of the Department of Molecular and Cell Biology at the University of California in Berkeley and colleages have engineered a new pathway for adding fluorine, and the new pathway can incorporate fluorine into smaller molecules than was possible before.
In a paper published in Nature the researchers describe how they used in vitro and in vivo methods to engineer the biochemical pathways usually used by soil bacteria (Streptomyces cattleya) to incorporate fluorine into building block molecules and then incorporate these into polyketides. The bacteria naturally catalyze the formation of carbon-fluoride bonds from aqueous fluoride and are therefore potentially a rich source of organic building blocks containing fluorine.
Polyketides are a large group of molecules that incorporate acetate into a wide range of biological molecules, and are important components of many insecticides, anti-fungal chemicals, and antibiotics. They are made naturally by some bacteria, plants and fungi to kill their bacterial or fungal predators. Beneficial polyketides are also found in red wine and green tea.
The results of the research take scientists a step closer to being able to insert fluorine into a much wider range of biological molecules that normally do not contain this element, and also enables them to insert fluorine into smaller biologically useful molecules than was previously possible.
More information: Expanding the Fluorine Chemistry of Living Systems Using Engineered Polyketide Synthase Pathways, Science 6 September 2013: Vol. 341 no. 6150 pp. 1089-1094 DOI: 10.1126/science.1242345
Abstract
Organofluorines represent a rapidly expanding proportion of molecules that are used in pharmaceuticals, diagnostics, agrochemicals, and materials. Despite the prevalence of fluorine in synthetic compounds, the known biological scope is limited to a single pathway that produces fluoroacetate. Here, we demonstrate that this pathway can be exploited as a source of fluorinated building blocks for introduction of fluorine into natural-product scaffolds. Specifically, we have constructed pathways involving two polyketide synthase systems, and we show that fluoroacetate can be used to incorporate fluorine into the polyketide backbone in vitro. We further show that fluorine can be inserted site-selectively and introduced into polyketide products in vivo. These results highlight the prospects for the production of complex fluorinated natural products using synthetic biology.
© 2013 Phys.org