Unique route to stem cells: Designer proteins developed to deliver stem cells
(Phys.org) —Researchers have developed a new method to produce stem cells using designed proteins. The new system is more precise and more natural than current techniques and the team believe it could be a more efficient and safer route to producing stem cells.
Stem cells have the potential to be used to replace dying or damaged cells with healthy cells. This repair could have wide-ranging uses in medicine such as organ replacement, bone replacement and treatment of neurodegenerative diseases. This study brings closer to realising the full potential of stem cell technology.
"We have gone down a completely different road to standard practices to produce stem cells from adult cells," says Dr Pentao Liu, senior author from the Wellcome Trust Sanger Institute. "Current techniques to reprogramme cells are inefficient and it's imperative to find other ways to create stem cells."
"We hope that our novel approach to reprogramming cells into stem cells will become a new and safer alternative to current practices."
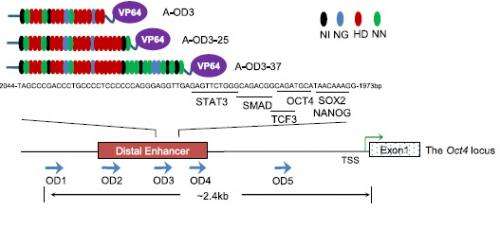
The team looked at proteins called transcription factors, which regulate the activity of all human genes. Each transcription factor acts to modify the activity of several or many genes.
A key set of these transcription factors are able to convert or reprogramme adult cells into induced pluripotent stem cells or iPS cells. However, these factors also act on many genes other than those involved in reprogramming.
The team developed artificial designer transcription factors to target those key reprogramming genes more accurately, minimizing activity on other genes.
"This is a promising and exciting development in our attempt to produce iPS cells that lend themselves in practical applications," says Dr Xuefei Gao, first author from the Wellcome Trust Sanger Institute. "We have shown that targeting gene-control regions, called enhancers, in this structured way is a very effective in controlling a gene and reprogramming cells to become iPS cells."
In conventional methods, the transcription factors used to programme cells take part in complicated ways - and target many different parts of the genome - as they are used to reprogramme the cells to become stems cells. As a result, the throughput of successfully reprogrammed cells can be low and the additional number of steps can have associated risks, such as affecting genes that can influence tumour development.
The designer transcription factors are extremely accurate. Because this method targets key genes directly and avoids additional genetic detours, it reduces the potential risks linked with standard practices.
The team looked at the structure of some unique transcription factors, called TALEs, and used a modular approach of taking the required components of designer transcription factors and stitching them together to make more precise and more specific factors that work only on the genes the team need to target. They hope this precision will lead to better and safer systems. The modular method should also make the development of new factors easier, speeding progress to a suite of stem cells for research and therapeutics.
Using designer transcription factors, the team successfully reprogrammed two cell types to become iPS cells; fibroblasts and EpiSCs. This method is also a promising way to ensure that stem cells divide and grow to form specific cell types such as heart cells, bone cells or brain cells.
"Stem cells hold enormous potential to help us understand and treat human disease, but currently there are gaps in our knowledge of how best to manipulate them," says Professor Allan Bradley, Director Emeritus of the Wellcome Trust Sanger Institute. "This is a unique and visionary way of bypassing many of the limitations currently associated with the manipulation of stem cells for both basic science and therapeutic applications."
More information: Gao, X. et al. Reprogramming to pluripotency using designer TALE transcription factors targeting enhancers, Stem Cell Reports, 2013. DOI: 10.1016/j.stemcr.2013.06.002
Journal information: Stem Cell Reports
Provided by Wellcome Trust Sanger Institute















