March 15, 2013 report
Microbes surviving deep inside oceanic crust
(Phys.org) —A new study shows for the first time that microorganisms are thriving deep within the oceanic crust under the sea floor, and hence far from light or oxygen.
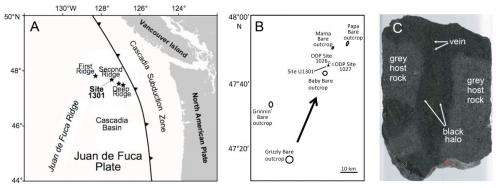
The paper, published in Science, reports on the analyses of samples of oceanic crust collected during a 2004 expedition to 100 kilometers off the Juan de Fuca Ridge off the coast of Washington state in the U.S..
The oceanic crust is a layer of volcanic rock several kilometers thick and covered by thick layers of sediments. Oceanic rock covers around 60 percent of the Earth's surface and is therefore potentially the largest biosphere on the planet. Microorganisms have been discovered on exposed surfaces of the oceanic crust and in the sediments above it, but the new study is the first to reveal that microbes also live deep within the crust.
Mark A. Lever, of the Department of BioScience at Aarhus University in Denmark and an international team of microbiologists drilled through the sea-bottom sediment and deep into the oceanic crust beneath to collect samples for various tests. The sediment depth at the drilling site was about 260 meters thick, and the team collected samples from about 350 to 580 meters deep into the basalt rock beneath, in a region known as the "dark biosphere." The age of the rock was estimated at 3.5 million years
The team ensured all their equipment was sterile to ensure the rock samples were uncontaminated. They also added marker chemicals to the fluid used in drilling, and later confirmed that while these chemicals were found on the rock sample surfaces, almost none found their way inside the rocks. They sterilized the outer surfaces before breaking the rock samples open.
The researchers found microbe genes within the rock samples, and to determine if the genes were from living microorganisms or extinct species, they incubated the samples at 65 degrees C (the temperature of the location at which they were found) in water similar chemically to that in their native location, which is rich in chemicals but poor in oxygen. After two years (enough for the slow-growing microorganisms to re-establish themselves) they transferred samples to another container containing sterilized rocks and the same water. They incubated these samples for a further five years.
The incubated samples began to produce methane and the carbon-13 concentration reduced, which showed that the microbes in the rock were alive. The scientists concluded the organisms were most likely deriving energy from chemical reactions taking place in the interfaces between the iron-laden rock and water. The chemical reactions produce hydrogen, which the microorganisms use to produce organic matter. Other microbes were identified, which survived by consuming sulfur. These processes of energy production are known as chemosynthesis.
The findings suggest that the ecosystems in the oceanic crust are fundamentally different from other ecosystems in that their energy is derived ultimately from chemosynthesis rather than photosynthesis, which derives energy from light.
Chemosynthesis is also known to be used by life forms in habitats such as deep boreholes or hydrothermal vents on the fringes of continental plates. The new study suggests that chemosynthesis is possibly more important than photosynthesis in terms of the biomass involved in the processes, depending on how extensive these sort of ecosystems are. Lever also said it is "quite likely there is similar life on other planets."
Dr Lever and his team expect to analyze other samples collected from ocean crusts beneath the North Atlantic and other locations in the Pacific Ocean.
More information: Evidence for Microbial Carbon and Sulfur Cycling in Deeply Buried Ridge Flank Basalt, Science 15 March 2013: Vol. 339 no. 6125 pp. 1305-1308 DOI: 10.1126/science.1229240
Abstract
Sediment-covered basalt on the flanks of mid-ocean ridges constitutes most of Earth's oceanic crust, but the composition and metabolic function of its microbial ecosystem are largely unknown. By drilling into 3.5-million-year-old subseafloor basalt, we demonstrated the presence of methane- and sulfur-cycling microbes on the eastern flank of the Juan de Fuca Ridge. Depth horizons with functional genes indicative of methane-cycling and sulfate-reducing microorganisms are enriched in solid-phase sulfur and total organic carbon, host δ13C- and δ34S-isotopic values with a biological imprint, and show clear signs of microbial activity when incubated in the laboratory. Downcore changes in carbon and sulfur cycling show discrete geochemical intervals with chemoautotrophic δ13C signatures locally attenuated by heterotrophic metabolism.
© 2013 Phys.org