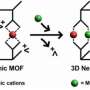
Ions shed water to slide into the perfect pore: Metal ions refuse to lose for large gaps, but will for a tighter fit

(Phys.org)—To slide into nano-sized openings inside minerals, certain metals shed layers of water, according to the NISE (pronounced "nice") theory, first published in 2011 by Dr. Cristian Schulthess and his colleagues. Recently, scientists at Pacific Northwest National Laboratory worked with Schulthess and his student Dan Ferreira to test the NISE theory. Ferreira traveled to PNNL, where he was assisted by Dr. Eric Walter and Dr. James Amonette in the use of a specialized instrument at EMSL that measures key aspects of a metal's behavior. They compared the reactions of manganese and copper ions with three different-sized pores in zeolite, a common type of mineral. Artwork from this study graced the cover of Clays and Clay Minerals.
"Usually charge wins over everything else," said Amonette, a PNNL geochemist. "However, in dealing with nanopores in zeolites, the sizes of the pores matter more."
While the behavior of certain charged metal particles, too small to see with the naked eye, may seem the stuff of ivory towers, it affects real-world problems, such as food crop yields and toxic waste dispersion. Scientists are determining what influences the behavior of these positively charged metal particles or metal cations. In most situations, the more highly charged the cation, the more readily it sticks to mineral surfaces. However, when the surfaces are located inside of nanopores, this rule doesn't always hold true. The NISE theory offers a clear explanation for this unusual result. This research gives further evidence that NISE accurately describes what's happening. The theory is a key step to understanding and then controlling the ions.
"Ions are constantly competing with each other and with bulk water molecules for retention sites on mineral surfaces (bind with them). The water molecules also wish to bind directly with the ions (hydrate them). This leads to many interesting field applications, such as the selective remediation of soil contaminants. But the rules of competition are radically different in these tight, claustrophobic environments that disrupt the stability of the hydrated ions," said Schulthess, an Associate Professor at the University of Connecticut.
This research project actually began at a Mexican restaurant in San Antonio, Texas. Schulthess and Amonette were attending a professional conference and talking about the NISE theory over dinner. Schulthess was continuing to test the theory with different metals and different instruments. He wanted to test the theory with calcium ions and a nuclear magnetic resonance (NMR) spectrometer, but the difficulty of accessing the specialized probes was proving problematic.
Amonette suggested an alternative. Test the theory with the electron paramagnetic resonance (EPR) spectrometer at the Environmental Molecular Sciences Laboratory using doubly charged manganese (Mn+2) and copper (Cu+2) ions. The instrument provides a wealth of data on the local environments of these metal ions. Ferreira and Schulthess submitted a user proposal that was accepted. Shortly thereafter, Ferreira was working with Amonette and Walter, the expert in EPR spectroscopy.
Using the EPR, the team examined manganese and copper and three different forms of nanoporous zeolite: zeolite Y with large pores, mordenite with small pores, and ZSM-5 with intermediate pores.
"Individually, people have done experiments with these zeolites and probably with test metals," said Walter. "But, they never did it all together. They never did the range of zeolite pores and the range of metals so that they could do a direct comparison and show the effect of pore size on metals."
Ferreira added, "We had used NMR to get direct spectroscopic evidence that singly charged ions were behaving the way the NISE theory said they should inside the zeolite nanopores. To complete that research though, we needed to confirm the predictions the NISE effect made about doubly charged ions. NMR was not feasible. Jim's suggestion to use EPR instead was a great idea because we could get very precise measurements of both manganese and copper in the zeolite nanopores. This was exactly the evidence we needed to complete our spectroscopic research."
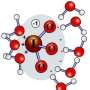
The team determined that the manganese and copper cations moved easily into the larger nanopores without losing their surrounding layers of water. The cations did not bind tightly to the mineral. Conversely, the cations shed their surrounding water molecules to move into the smallest pores, bonding with the mineral. However, when it came to the intermediate-sized pores, the manganese and copper behaved differently. The manganese kept its water layer and bonded inside the pores. The copper ions shed their water and bonded tightly with the mineral, although not as tightly as they bonded in the smaller pores. This study showed that the physical structure of the retention sites can strongly influence chemical processes and the strength of retention.
More information: Ferreira, D. et al. 2012. An Electron Paramagnetic Resonance Spectroscopy Investigation of the Retention Mechanisms of Mn and Cu in the Nanopore Channels of Three Zeolite Minerals. Clays and Clay Minerals 60(6):588-598. DOI: 10.1346/CCMN.2012.0600604
Provided by Pacific Northwest National Laboratory