Chemical probe finds fungal organism function: Activity-based protein profiling suggests how fungus becomes pathogenic
(Phys.org)—Two unique chemical probes designed at Pacific Northwest National Laboratory are helping scientists find how a pathogenic organism responsible for a severe lung infection thrives in human serum. These probes use multiplexed activity-based protein profiling (ABPP), which revealed significant changes in Aspergillus fumigatus metabolism and stress response when placed in culture with human serum over time.
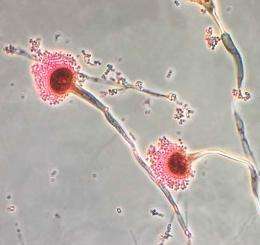
A. fumigatus spores are ubiquitous in the atmosphere, and everybody inhales an estimated several hundred spores each day. But for people whose immune response is compromised from taking immunosuppressive drugs, by irradiation or malnutrition, or by diseases such as cancer or AIDS, this opportunistic pathogen is responsible for pulmonary invasive aspergillosis (IA). Patients with IA are usually critically ill, and the disease is difficult to cure.
The PNNL team hypothesized that A. fumigatus uses human serum, the clear fluid that remains after blood is allowed to clot, as a nutrient and that enzyme activity within the serum impacts the organism's metabolism, nutrient sensing, and scavenging response within an immunocompromised host environment. They simultaneously used two novel activity-based chemical probes they designed and constructed to target the reactivity of A. fumigatus during growth. The team found that probe-protein reactivity changes in the presence or absence of human serum.
The information provides valuable insight into how A. fumigatus survives in a host environment on a fundamental level. During the course of IA, the fungus' filamentous structure breaches host tissue and interacts with serum, where it readily grows because of its unique ability to extract iron from human transferrin in an iron-limited environment. Furthermore, A. fumigatus can use serum proteins as building blocks for growth, but the full effect of serum on its cellular processes and its relevance to disease are not fully understood.
"We are demonstrating that we can compare two systems or one organism under multiple conditions to tease out more information about protein regulation," said Dr. Susan Wiedner, a PNNL Linus Pauling Distinguished Postdoctoral Fellow, and lead author of the paper published in The Journal of Biological Chemistry.
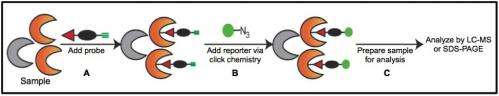
The scientists wanted to determine which proteins interact with a small-molecule activity-based probe. "We found that under two different growth conditions, the number and identity of proteins that interact with the probes change drastically," said Wiedner. "We could measure the abundance of probe-labeled proteins by liquid chromatography-mass spectrometry (LC-MS) -based proteomics and see profiles of labeled proteins based on growth condition."
Various approaches to LC-MS-based proteomics have emerged. In a typical global analysis, thousands of proteins are measured from a complex proteome. However, by using ABPP, scientists can target a subset of a few hundred proteins from the complex proteome.
"ABPP is a more directed approach than using global proteomics," said Wiedner. "We can look at the difference of probe-labeled protein abundances among various systems and conditions. This then tells us more about the system's biology, such as the differences between probe-reactivity of metabolic proteins. Some fungal proteins interact more with the probe in the presence of human serum, which tells us something about what metabolism might be doing under those conditions." A direct comparison of a global analysis and an ABPP analysis showed differences in measured proteins detected by ABPP that were not detected by global analysis.
In turn, this can lead to more in-depth studies such as generating gene knock-out mutants and performing enzymatic assays, all of which could be used to develop effective treatments and detection of IA.
Over the last two decades, ABPP development has been a growing but still-small field. PNNL has one of the groups working on this, led by PNNL chemist Dr. Aaron Wright, senior author on this paper.
Currently, PNNL scientists are developing ABPP probes and supporting bioinformatics capabilities to measure cellulose degradation in microbial communities. ABPP can be useful for a variety of things including target validation of drug candidates and protein inhibitor discovery.
Said Wright, "Probes can compete with known drugs for protein-binding sites, which results in drug target and drug off-target validation. Some probes are broad, like those used in this study. But some can be very selective for an enzyme class. We can design the probe based on the enzyme class being targeted. We used a multiplexed approach here, where two probes were used simultaneously to target more than one type of protein. Previous studies only use one ABP at a time for proteome analysis."
More information: Wiedner, S. et al., Multiplexed Activity-based Protein Profiling of the Human Pathogen Aspergillus fumigatus Reveals Large Functional Changes upon Exposure to Human Serum. The Journal of Biological Chemistry, 2012, 287(40):33447-33459. DOI: 10.1074/jbc.M112.394106.
Journal information: Journal of Biological Chemistry
Provided by Pacific Northwest National Laboratory