September 3, 2012 report
Researchers devise simple and cheap method to detect TB in patients
(Phys.org)—Tuberculosis or TB as it's become more commonly known, is a horrible disease by all accounts, it slowly kills many of its victims, particularly those living in the developing world. In 2010, it killed an estimated four thousand people every single day, which is particularly horrendous when noting that many of those who succumb to its effects could be have been saved were they to be diagnosed and treated in a timely manner. Unfortunately, in many areas of the world neither is available, thus the news that a team of researchers working together from several universities in the US has developed a new kind of test that reveals the presence of TB in patients, both quickly and cheaply, is truly exciting. The new probe, as the team describes in their paper published in Nature Chemistry, can allow for TB detection using nothing more than a simple box housing light emitting diodes and some filters.
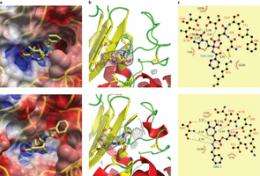
The probe is based on BlaC, a protein that TB produces and its impact on β-lactams, which are a certain class of chemicals. Normally, BlaC simply breaks them down. With this new probe however the team created a modified version of a β-lactam that is cut or cleaved by BlaC, rather than being broken down by it haphazardly. But that's only the beginning, they've also inserted a fluorescent molecule into the probe, allowing it to be seen when cleaved by TB. The test is very simple, if the fluorescent molecule can be seen (using the LED box and filters) in a sample of sputum, then TB must be present to make it so. The faint light produced is enough to be seen in images taken by a simple camera in a phone and shown to clinicians.
Up till now, the only way to test for TB in remote patients was to collect a sputum sample and send it to a location that had a microscope, where trained clinicians looked for the TB bacteria. Sadly, this method is not only slow, it's also relatively inaccurate when there are few bacteria to be seen, such as is the case with infected children. With the new probe, test times can be reduced to mere minutes and accuracy is improved dramatically.
Reducing the amount of time it takes to test for TB not only helps the patient, it helps those around them too because TB is of course, communicable, with some estimating that one infected, untreated person may account for as many as ten or fifteen new infections in others in just one year.
More information: Rapid point-of-care detection of the tuberculosis pathogen using a BlaC-specific fluorogenic probe, Nature Chemistry (2012) doi:10.1038/nchem.1435
Abstract
Early diagnosis of tuberculosis can dramatically reduce both its transmission and the associated death rate. The extremely slow growth rate of the causative pathogen, Mycobacterium tuberculosis (Mtb), however, makes this challenging at the point of care, particularly in resource-limited settings. Here we report the use of BlaC (an enzyme naturally expressed/secreted by tubercle bacilli) as a marker and the design of BlaC-specific fluorogenic substrates as probes for Mtb detection. These probes showed an enhancement by 100–200 times in fluorescence emission on BlaC activation and a greater than 1,000-fold selectivity for BlaC over TEM-1 β-lactamase, an important factor in reducing false-positive diagnoses. Insight into the BlaC specificity was revealed by successful co-crystallization of the probe/enzyme mutant complex. A refined green fluorescent probe (CDG-OMe) enabled the successful detection of live pathogen in less than ten minutes, even in unprocessed human sputum. This system offers the opportunity for the rapid, accurate detection of very low numbers of Mtb for the clinical diagnosis of tuberculosis in sputum and other specimens.
Journal information: Nature Chemistry
© 2012 Phys.org















