Worms can evolve to survive intersex populations

(PhysOrg.com) -- Sexually reproducing species need at least two sexes in order to produce offspring, but there are many ways that nature produces different sexes. Many animals (including humans and other mammals) use a chromosomal sex determination system in which females have two X chromosomes, while males have one X and one Y.
On the other hand, for some reptiles, sex is temperature dependent--that is, incubating the eggs at different temperatures can determine whether the offspring develop as males or females.
Evolution in a trait can only happen when variation exists in that trait. However, gene mutations related to sex determination could be very risky if they result in animals that are unable to reproduce.
Evolution of sex
Researchers have long wondered how variation in such a trait could exist, since we might expect sex determination to be highly conserved--meaning that any variation in the genes would probably lead to trouble and thus be quickly eliminated by natural selection. To the contrary, there is evidence that these genes have evolved very rapidly and diverged greatly among species. But what happens in the intermediate evolutionary stages of sex determination?
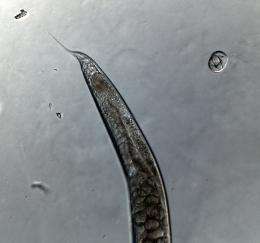
Under normal circumstances, the worm-like nematode Caenorhabditis elegans (C. elegans) has a chromosomal sex determination system: most individuals are XX, which are actually hermaphrodites that can self-fertilize, but can also outcross with males, who only have a single X chromosome and develop male gonads. In a new paper in the journal Evolution by Michigan State University postdoctoral researcher Christopher Chandler and assistant professor Ian Dworkin--both members of the BEACON Center for the Study of Evolution in Action--along with collaborators at Iowa State and the University of Oregon used experimental evolution to test how C. elegans could overcome changes to that system.
The researchers used a mutant strain of C. elegans that has a temperature-dependent sex determination system: At cool temperatures, the animals typically develop as hermaphrodites, but if they are exposed to high temperatures during development, they become males. The researchers produced populations full of intersex individuals by exposing the larvae to intermediate temperatures. The intersex individuals had characteristics of both males and females, which made reproduction difficult for them.
Intersex reproduction
Chandler and colleagues then allowed these low-fertility, intersexed populations to survive and reproduce for 50 generations. At that point, they measured the later generations' sex ratio and fertility.
C. elegans sexes can be easily distinguished by tail shape: Males have a rounded tail tip, while females have a whip-like, pointed tip and the eggs can easily be seen near the tip of the tail. The intersex animals have both characteristics, with a rounded male-like tip but often carrying eggs like a female.

By the end of the experiment, all of the populations had returned to more typical sex ratios and higher fertility, despite being subjected to the intermediate temperature that originally produced intersex animals. The researchers found that the animals had not changed much in the expression of sex determination genes. Instead, Chandler said, "they appeared to be compensating through other genetic mechanisms, and likely in a number of different ways."
In other words, other genes were evolving to make up for changes in the sex determination genes, in a way that allowed individual animals to develop either as a male or a female, rather than intersex.
The findings have enormous implications for understanding how evolution works. As Dworkin points out, "even when the genes have a function as crucial as sex determination, the evolutionary process can overcome intermediate or less-fit types in sometimes surprising ways."
Provided by National Science Foundation















